Translational Biophotonics in Developmental Disorders and Diseases

- Amir H. Gandjbakhche,
PhD, Head, Section on Translational Biophotonics - Thien Nguyen, PhD, Research Fellow
- Maria Ayoub, PhD, Postdoctoral Fellow
- Tanya Azar, MD, Postdoctoral Fellow
- Jinho Park, PhD, Postdoctoral Fellow
- Soo Yoong Park, PhD, Postdoctoral Fellow
- Wan-Chun Su, PhD, Postdoctoral Fellow
- Marc Bornstein, PhD, Special Volunteer
- Wei Lun Huang, MS, Intramural Research Training Award Student
- Minghao Xue, BS, Intramural Research Training Award Student
- Cansu Erkan, BS, Postbaccalaureate Fellow
- Abigail Frankenberg, BS, Postbaccalaureate Fellow
- Aarani Kamalanathan, BS, Postbaccalaureate Fellow
- John Mutersbaugh, BS, Postbaccalaureate Fellow
- Asma Sodager, BS, Postbaccalaureate Fellow
Brain imaging and spectroscopy of chronic developmental disorders
We are using fNIRS (functional near-infrared spectroscopy), EEG (electroencephalography), and Deep Learning models to study chronic developmental disorders such as ASD (autism spectrum disorder), DMDD (disruptive mood dysregulation disorder), and PCI (parent child interaction). We aim to comprehensively explore typical and atypical developmental trajectories in infants and children, including those at elevated risk for developmental disorders. Such research efforts will help us to provide more accurate insights into neural mechanisms through multi-modal imaging, and identify potential neuro-biomarkers for children with developmental disabilities.
To characterize the MNN (mirror neuron network) using EEG and fNIRS, we developed a multimodal data-fusion approach that leverages EEG’s temporal and fNIRS’s spatial resolution. We used Mu suppression as a marker of MNN activation in EEG data. Using ssmCCA (structured sparse multiset canonical correlation analysis), we identified congruent activity during action execution and observation across key left-hemisphere regions, including the paracentral, precentral, and parietal cortices.
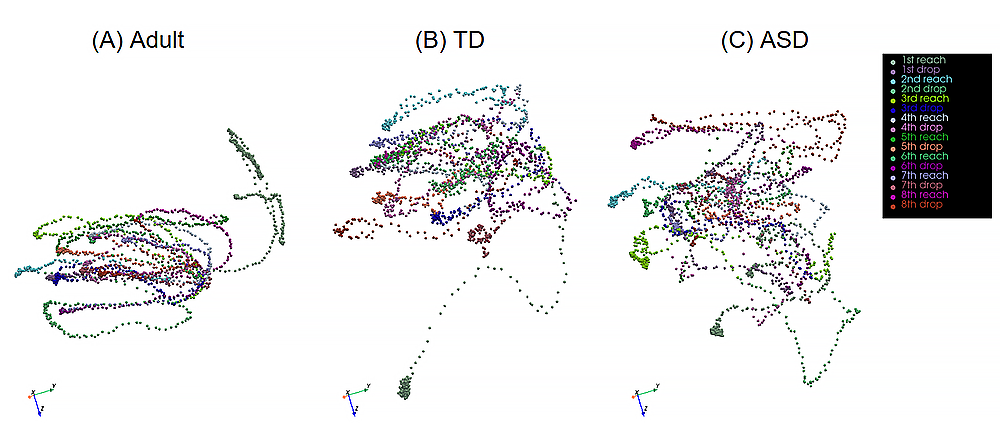
It is known that ASD is among the most prevalent neurodevelopmental disorders, yet the current diagnostic procedures rely on behavioral analyses and interviews, without objective screening methods to support the diagnostic process. To address this gap, in collaboration with Anjana Bhat and Wan-Chun Su, a former postdoctoral fellow in our Section, we integrated upper-limb kinematics and deep-learning methods to identify potential biomarkers that, in the future, could be validated in younger age groups to enhance the identification of ASD. Forty-one school-age children, with and without an ASD diagnosis (mean age ± SE: TD (typically developing) group: 10.3 ± 0.8, 8 males and 7 females; ASD group: 10.3 ± 0.5, 21 males and 5 females), participated in the study (Figure 1). A single IMU (inertial measurement unit) was affixed to the child’s wrist as they engaged in a continuous reaching and placing task (Figure 2). We employed deep learning techniques to classify children with and without ASD [Reference 1]. Our findings suggest differential movement kinematics in school-age children compared with healthy adults. Compared with TD children, children with ASD exhibited poor feedforward/feedback control of arm movements as seen by a greater number of movement units, more movement overshooting, and prolonged time-to-peak velocity/acceleration. Unique movement strategies such as greater velocity and acceleration were also seen in the ASD group. More importantly, using a MLP (multilayer perceptron, an artificial neural network consisting of multiple layers of neurons) model, we demonstrated an accuracy of about 78.1% in classifying children with and without ASD. These findings underscore the potential use of studying upper-limb movement kinematics during goal-directed arm movements and deep-learning methods as valuable tools for classifying and, consequently, aiding in the diagnosis and early identification of ASD, upon further validation of their specificity among younger children [Reference 1]. We then assessed a variety of deep-learning approaches for the classification of ASD. The best result was achieved with a convolutional autoencoder combined with long short-term memory layers, reaching an accuracy of 90.21% and an F1-score of 90.02%. Once the convolutional autoencoder together with long short-term memory was determined to be the most effective model for this datatype, it was retrained and evaluated with a patient-separated dataset to assess the generalization capability of the model, achieving an accuracy of 91.87% and an F1-score of 93.66% [Reference 2].
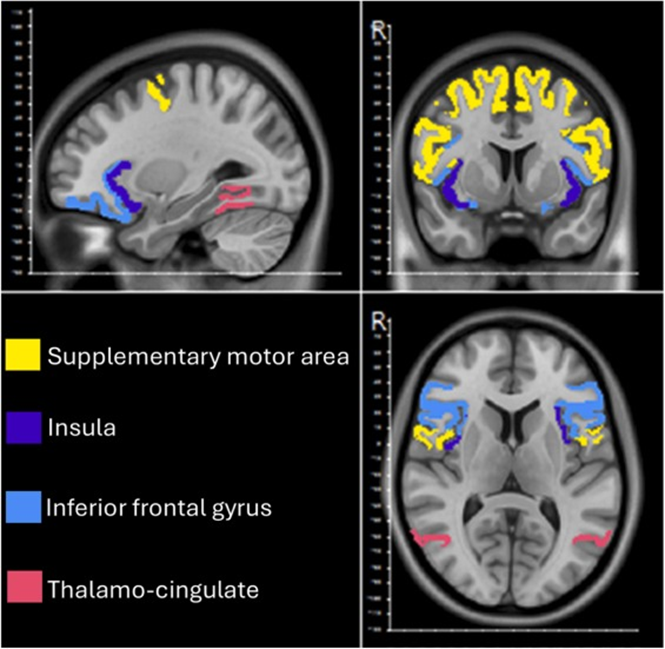
In collaboration with Marc Bornstein, we studied parent brain responses to child facial and vocal communication [Reference 3]. Parenting consists of caregiving cognitions and practices, based in evolution and biology as well as culture and context, and lodged in nervous-system structure and function. This narrative review first discusses parenting and an orientation toward caregiving in terms of its nature, constituents, and goals. Next, the review operationalizes parenting cognitions and practices that are expressed in response to facial and vocal cues of young offspring and revealed in naturalistic and experimental studies using behavioral preferences, attention tasks, and implicit association tests. We subsequently reviewed studies of autonomic and central-nervous-system structures and functions in the service of parenting (Figure 3). Taken together, these investigations of the parent nervous system reveal associations with, and likely neuropsychological underpinnings of, parenting cognitions and practices and begin to unveil specific mechanisms and loci in the human nervous system that define a neuroscience of parenting.
Figure 3. Brain regions involved in responses to infant facial communication (brain images generated from DSI Studio, a tractography software tool)
Activity map of brain regions involved in responses to infant facial communication; regions such as insula, inferior frontal gyrus, and thalamo-cingulate, along with motor exhibit larger activity
Figure 3. Brain regions involved in responses to infant facial communication (brain images generated from DSI Studio, a tractography software tool)
Activity map of brain regions involved in responses to infant facial communication; regions such as insula, inferior frontal gyrus, and thalamo-cingulate, along with motor exhibit larger activity
We are expanding an existing NIMH protocol (02-M-0021) to incorporate fNIRS as an additional objective measure to better characterize irritability and treatment response in children and adolescents with DMDD. Without altering its core design or methodology, this amendment builds upon the current protocol by integrating a noninvasive, portable neuroimaging approach alongside established behavioral assessments. DMDD is characterized by persistent irritability and severe temper outbursts that significantly impair daily functioning, yet the neural mechanisms underlying emotion dysregulation remain poorly understood. Using fNIRS, we aim to measure prefrontal cortical activation patterns associated with emotion regulation at rest and during task-based paradigms before, during, and after a 12-week course of CBT (cognitive behavioral therapy). Participants will complete the Flanker and Stop-Signal tasks to probe cognitive control, inhibitory processing, and emotional regulation, which are domains closely linked to irritability in DMDD. This child-friendly, multimodal approach is designed to provide objective neural markers of treatment response, improve understanding of the neurobiological basis of irritability, and support the development of more precise outcome measures for pediatric psychiatric interventions.
Wireless multimodal point-of-care biosensor to study respiratory disorders
Using NIRS, we developed a multimodal biosensor, which can measure chest-tissue oxygenation levels as well as cardiac and respiratory functions. The biosensor is currently being used for two different studies. The first study is to monitor participants with different breathing patterns, and the second is to monitor pediatric patients with obstructive sleep apnea. For the first study, we developed TCNN (a two-stream convolutional neural network) for breathing pattern classification [Park J, et al., Bioengineering (Basel) 2024;11:709]. The TCNN consists of a CNN–based autoencoder and classifier. The encoder of the autoencoder generates deep compressed feature maps, which contain the most important information-constituting data. These maps are concatenated with feature maps generated by the classifier to classify breathing patterns. We applied the TCNN, SCNN (single-stream CNN), and state-of-the-art classification models to classify four breathing patterns: normal, slow, rapid, and breath holding. The input data consisted of chest-tissue hemodynamic responses measured using a wearable near-infrared spectroscopy device on 14 healthy adult participants. Among the classification models evaluated, random forest, a machine-learning algorithm, had the lowest classification accuracy at 88.49%, while the TCNN achieved the highest classification accuracy at 94.63%. In addition, the proposed TCNN performed 2.6% better in terms of classification accuracy than an SCNN (without an autoencoder). Moreover, the TCNN mitigates the issue of declining learning performance with increasing network depth, as observed in the SCNN model. These results prove the robustness of the TCNN in classifying breathing patterns despite using a significantly smaller number of parameters and computations compared with state-of-the-art classification models.
For the second study, we developed a wearable device designed to detect sleep apnea in children, specifically targeting the challenges of accurately monitoring oxygen saturation during sleep. This wireless NIRS device measures tissue oxygen levels, heart rate, and breathing patterns noninvasively, offering a more reliable alternative to traditional pulse oximeters, which can be inaccurate in certain populations. Currently, we are collaborating with Ashura Buckley and Bruce Tromberg to test this device on pediatric patients with and without obstructive sleep apnea (NICHD protocol # 000224). The goal is to measure chest-tissue oxygen saturation during sleep apnea events and validate the results against clinical data. The device could improve the diagnosis and monitoring of OSA (obstructive sleep apnea) in children, offering a less invasive, more precise method for tracking the physiological impacts of OSA and guiding better treatment decisions. In addition, we conducted a systematic review to explore the effects of sleep apnea on children's psychological, developmental, and cognitive outcomes. Sleep apnea, often caused by obstructive airway issues such as enlarged tonsils, disrupts sleep patterns and oxygen levels, impairing brain development. Children with untreated sleep apnea are at higher risk for learning difficulties, behavioral issues, and poor academic performance. Our review aimed to fill gaps in the existing literature by examining these combined effects, and we conducted a meta-analysis to synthesize the findings. These suggest a potential link between pediatric obstructive sleep apnea and adverse cognitive (overall effect size = –0.73 [–1.11, –0.36] for verbal WISC [Wechsler Intelligence Scale for Children]), psychological (overall effect size = 2.26 [–0.29, 4.81] for total CBCL [child behavior checklist]), and developmental (overall effect size = 1.90 [0.13, 3.67] for global BRIEF [Behavior Rating Inventory of Executive Functioning]) outcomes. These results may help inform recommendations for early screening and intervention. Following this, we plan original research involving children with sleep apnea to assess these outcomes and inform clinical practices.
Multimodal point-of-care biosensor for monitoring pregnancy health
Placental oxygenation
Poor placental development and placental defects can lead to adverse pregnancy outcomes such as pre-eclampsia, fetal growth restriction, and stillbirth. We developed two sensors, which use a NIRS technique to measure placental oxygen saturation transabdominally [Nguyen T, et al., Biosensors (Basel) 2024;14:481]. The first one, an NIRS sensor, is a wearable device consisting of multiple NIRS channels. The second one, a multimodal sensor, which is an upgraded version of the NIRS sensor, is a wireless and wearable device, integrating a motion sensor and multiple NIRS channels. We conducted a pilot clinical study to assess the feasibility of the two sensors in measuring transabdominal placental oxygenation in 36 pregnant women (n = 12 for the NIRS sensor and n = 24 for the Multimodal sensor) at the Center for Advanced Obstetrical Care and Research of the Perinatology Research Branch, located at the Detroit Medical Center, under protocol #090717MP4E. Among these subjects, four participants had an uncomplicated pregnancy, and 32 patients had either maternal pre-existing conditions/complications, neonatal complications, and/or placental pathologic abnormalities. The study results indicate that the patients with maternal complicated conditions (69.5 ± 5.4%), placental pathologic abnormalities (69.4 ± 4.9%), and neonatal complications (68.0 ± 5.1%) had statistically significantly lower transabdominal placental oxygenation levels than those with an uncomplicated pregnancy (76.0 ± 4.4%; F [3,104] = 6.6, p = 0.0004). Additionally, the study demonstrates the capability of the Multimodal sensor for detecting the maternal heart rate and respiratory rate, fetal movements, and uterine contractions. The findings demonstrate the feasibility of the two sensors in the real-time continuous monitoring of transabdominal placental oxygenation to detect at-risk pregnancies and guide timely clinical interventions, thereby improving pregnancy outcomes.
On this topic, we conducted a review of antenatal fetal monitoring techniques [Reference 4] that can help patients and healthcare providers recognize and intervene in a timely manner to prevent conditions that threaten fetal well-being. We explored various fetal well-being assessment methods. The most used techniques include the non-stress test, contraction stress test, and biophysical profiles that encompass both fetal movement and fetal heart rate. The review delves into technologies used for antenatal diagnostic methods, ranging from traditional methods to contemporary advancements. The presented categories serve as a guide, recommending techniques tailored to specific pregnancy stages and observational purposes.
Dynamic full-field optical coherence tomography (DFFOCT)
In order to evaluate data collected from in vivo measurement, we have been assessing intrinsic intracellular activities of cells at different oxygen levels using the DFFOCT system. In our experimentation, we employ HeLa and HEK cells, which are similar to placental cells, so that the results of the study can be widely applied. We successfully constructed a customized incubator for cell cultivation in conjunction with the DFFOCT system. The incubator addresses past limitations associated with maintaining optimal temperature, gas concentration control, and the continuous supply of nutrients during extended cell observations using the DFFOCT system. Notably, the incubator's systematic control over the supplied oxygen concentration permits a more methodical exploration of the impact of placental oxygen levels on fetal development. We found that cell viability is highly subjective to temperature variations (22°C and 36°C) in our preliminary 24–hour longitudinal studies. During hypothermia, cells showed blebbing and swelling and decomposed into small fragments, resulting in cell death. Conversely, at body temperature, cell proliferation and intracellular dynamic movements were seen even after 24 hours. Intracellular activities were shown by DFFOCT in uniform movement with low mean frequency and high magnitude. We have filed a provisional patent application entitled “Dynamic Assessment of Cellular Metabolism Through Optical Imaging and Artificial Intelligence Techniques.”
Total body photography (TBF) for skin cancer research
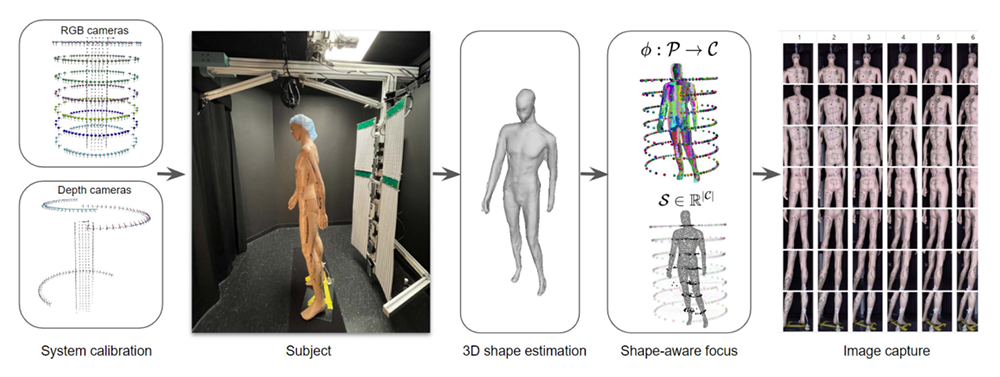
TBP is gaining popularity as a powerful tool for early melanoma detection. TBP allows monitoring of temporal changes in skin lesions, efficient screening of numerous lesions, and provides essential anatomical locations for dermatoscopic images. Despite its potential, several challenges are hindering its widespread application in skin cancer research. We propose a novel framework combining geometric and texture information to localize skin-lesion correspondence from a source scan to a target scan in TBP. We evaluated the framework quantitatively on both a public and a private dataset, for which our success rates (at 10 mm criterion) are comparable to the only reported longitudinal study [Huang W-L, et al., ArXiv 2023:arXiv:2307.09642v2;preprint], which we published. We formulated the image-capture problem as limited to the depth of field of a camera and designed a novel algorithm to improve overall image quality. The proposed EM (expectation-maximization) and k-view algorithms improve the relative cost of the baseline single-view methods by at least 24% and 28% respectively, corresponding to increasing the in-focus surface area in total body photography by roughly 1550 cm2 and 1780 cm2 [Huang W-L, et al., 2024, submitted to the Winter Conference on Applications of Computer Vision 2025]. In collaboration with Mehran Armand and Jun Kang, we developed a novel shape-aware TBP system for high-resolution, quality-surface-coverage-maximized, and multi-spectral images. A clinical protocol has been approved by the Johns Hopkins University institutional review board, and allows the study to evaluate the clinical effectiveness of the proposed system, with 12 patients collected.
Figure 4. Total body photography for skin cancer detection
The proposed imaging pipeline for a novel shape-aware total body photography system, including system calibration, 3D shape estimation, in-focus surface optimization, and image capture: we demonstrated that the proposed shape-aware focus outperforms existing focus protocols (i.e., auto-focus).
Figure 4. Total body photography for skin cancer detection
The proposed imaging pipeline for a novel shape-aware total body photography system, including system calibration, 3D shape estimation, in-focus surface optimization, and image capture: we demonstrated that the proposed shape-aware focus outperforms existing focus protocols (i.e., auto-focus).
Additional Funding
- Bench to Bedside Award 345 (2016): “Mirror neuron network dysfunction as an early biomarker of neurodevelopment” (ongoing)
- Human Placenta Project–NICHD (2016, ongoing)
- Scientific Director's Award
Publications
- Using deep learning to classify developmental differences in reaching and placing movements in children with and without autism spectrum disorder. Sci Rep 2024 14(1):30283
- Deep learning approaches for classifying children with and without autism spectrum disorder using inertial measurement unit hand tracking data: comparative study. JMIR Med Inform 2025 13:e73440
- Parent brain responses to child facial and vocal communication: What different methodologies reveal about parenting cognitions and practices as well as their moderation by parental status, gender, and culture as well as child characteristics. Neurosci Res 2025 218:104229
- From traditional to cutting-edge: a review of fetal well-being assessment techniques. IEEE Trans Biomed Eng 2025 72(5):1542-1552
- Shape-aware total body photography system for in-focus surface coverage optimization. IEEE J Biomed Health Inform 2025 29(10):7434-7445
Collaborators
- Franck Amyot, PhD, Center for Neuroscience and Regenerative Medicine, Uniformed Services University of the Health Sciences (USUHS), Bethesda, MD
- Mehran Armand, PhD, The Johns Hopkins University, Baltimore, MD
- Anjana Bhat, PhD, University of Delaware, Newark, DE
- Claude Boccara, PhD, École Supérieure de Physique et de Chimie Industrielles, Paris, France
- Melissa Brotman, PhD, Neuroscience and Novel Therapeutics Unit, NIMH, Bethesda, MD
- Ashura Buckley, MD, Sleep Disorders and Neurodevelopment Service, NIMH, Bethesda, MD
- Jun Kang, MD, The Johns Hopkins University, Baltimore, MD
- Jay Knutson, PhD, Laboratory of Molecular Biophysics, NHLBI, Bethesda, MD
- Guoyan Luo, MD, PhD, Howard University Hospital, Washington, DC
- Tom Pohida, MS, Division of Computational Bioscience, CIT, NIH, Bethesda, MD
- Randall Pursley, MSc, Signal Processing and Instrumentation Section, CIT, NIH, Bethesda, MD
- Roberto Romero-Galue, MD, D(Med)Sci, Perinatology Research Branch, NICHD, Detroit, MI
- Babak Shadgan, MD, MSc, PhD, University of British Columbia, Vancouver, Canada
- Wan-Chun Su, PhD, Louisiana State University, Baton Rouge, LA
- Audrey Thurm, PhD, Pediatrics & Developmental Neuropsychiatry Branch, NIMH, Bethesda, MD
- Bruce Tromberg, PhD, Section on Biomedical Optics, NICHD, Bethesda, MD
- Eric Wassermann, MD, Cognitive Neuroscience Section, NINDS, Bethesda, MD
Contact
For more information, email gandjbaa@mail.nih.gov or visit https://www.nichd.nih.gov/research/atNICHD/Investigators/gandjbakhche.