Innovative Approaches to Clinical Management of Complex Pregnancy Issues

- Katherine L. Grantz, MD, MS, Senior Investigator, Epidemiology Branch, DiPHR
- Jessica Gleason, PhD, Staff Scientist
- Sarah Thornburg, PhD, Postdoctoral Intramural Research Training Award Fellow
- Elizabeth Williams, PhD, Postdoctoral Intramural Research Training Award Fellow
- Virali Patel, BS, NIH Medical Research Scholars Program Fellow
Our research aims to improve care for pregnant women and their babies by addressing key challenges in pregnancy management, including abnormal fetal growth, determining the safest timing for delivery in high‑risk pregnancies, and optimizing labor and delivery practices. Findings from this work have shaped national and international clinical practice guidelines.
Novel measures of fetal growth and development: state-of-the-art tools to assess fetal health
Normal fetal growth is a marker of an optimal intrauterine environment and is important for long-term offspring health. Fetal size is monitored during pregnancy because fetal growth restriction and small-for-gestational-age (SGA) as macrosomia and large-for-gestational-age (LGA) are associated with increased risk of perinatal morbidity and mortality. Yet distinguishing fetal growth that is constitutionally small or large from growth that is pathologically restricted or increased presents a significant challenge in obstetrics. Cross-sectional fetal measurements are typically compared with reference size-for-age curves, with a range of 10th–90th percentiles considered appropriate for gestational age. Yet, a single measurement can only indicate size. At least two measurements separated in time are needed to estimate actual fetal growth. Fetal growth velocity is the rate of fetal growth over a given time interval (e.g., g per week). Understanding whether fetal growth has deviated from a normal trajectory may have more clinical utility in distinguishing between constitutional and pathologic fetal growth than using a particular threshold of fetal size based on a single time measure. Fetal growth, along with accurate gestational dating, are key factors in clinical decision-making for antenatal monitoring and determining the route and timing of delivery. Yet, until recently, there has been a lack of longitudinal prospective studies with diverse populations that have collected repeated ultrasound measurements. In addition, errors in ultrasound estimated fetal weight (EFW) prediction of birth weight have clinical implications such as timing of delivery for fetal undergrowth or decision for cesarean due to suspected macrosomia. Our research program addresses these critical data gaps using several approaches, including developing and applying fetal growth standards for use in clinical practice, investigating the role of fetal growth velocity, customized and individualized fetal growth references, and fetal 3D volumes.
The NICHD Fetal Growth Studies team was responsible for a multidisciplinary effort that generated fetal growth percentile charts in the U.S. population for clinical practice [Buck Lewis, et al. Am J Obstet Gynecol 2015;213:449; Grantz, et al. Am J Obstet Gynecol 2022;226:576]. Our team developed the first ever fetal growth velocity calculator for clinical use [Grantz, et al. Am J Obstet Gynecol 2018;219:28; Grantz, et al. Am J Obstet Gynecol 2022;227:916]. This past year, we followed up on this work and found that integrating fetal growth velocity into a birthweight-predicting formula resulted in small improvements in the prediction of birthweight, SGA, and LGA [Reference 2]. Given the clinical implications of misclassifying a fetus as SGA or LGA at birth, even small improvements in the prediction of body weight by incorporating fetal growth velocity may translate into improved prenatal care by avoiding unnecessary interventions and associated morbidity on a population level. The benefits of using growth velocity to categorize fetal growth and assess its contribution to birth weight had not been empirically demonstrated prior to our work in this area.
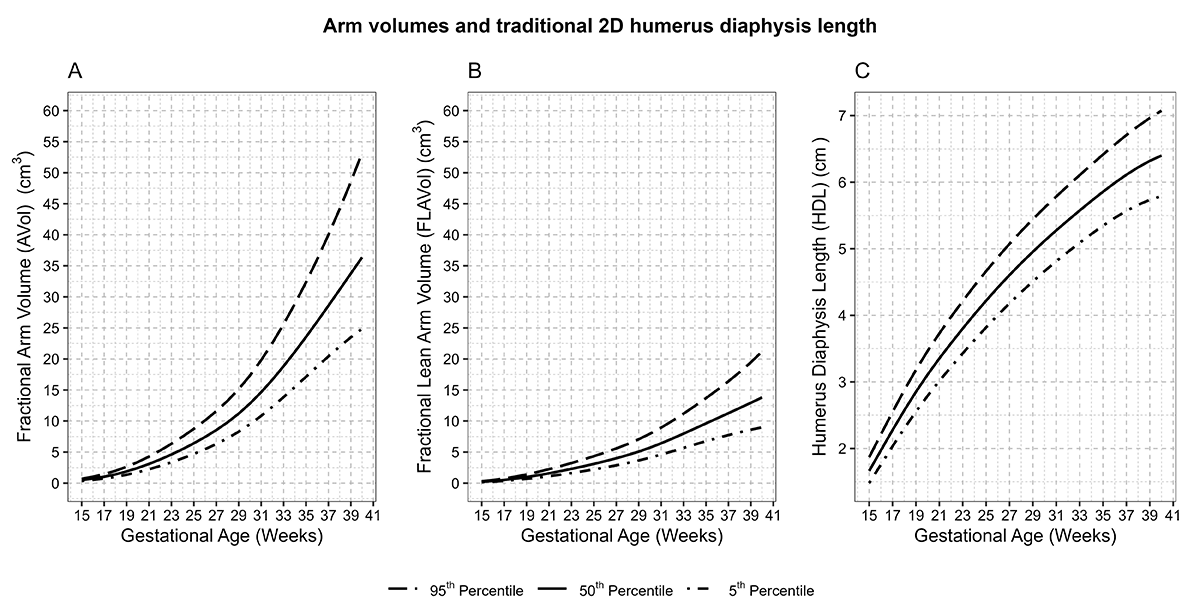
Our group is among the first to have accumulated the largest collection of fetal 3-dimensional (3D) volumes from a heterogeneous pregnancy cohort with repeat ultrasounds spanning the length of gestation. While 3D ultrasound capability is generally available, it has not been widely adopted into clinical practice. This past year, we established novel standards for fetal soft-tissue and organ-volume measurements by 3D ultrasound technology [Reference 3]. Notably, growth patterns and timing of maximal growth for 3D lean and fat measures, and limb and organ volumes differed from patterns revealed by traditional 2D growth measures. For example, fractional arm and lean arm volumes, depicted in panels A and B of Figure 1, increased with acceleration around 29–30 weeks. In contrast, the 2D humerus diaphysis length demonstrated a logarithmic growth curve, with the fastest growth from 16–26 weeks. These findings suggest that novel 3D fetal growth parameters reflect unique facets of fetal growth. Based on these findings, we are now investigating whether growth in these 3D measures may be altered by genetic, nutritional, metabolic, or environmental influences and pregnancy complications, in ways not identifiable using corresponding 2D measures.
Figure 1. Arm volumes and traditional 2D humerus diaphysis length
Distribution of A: Fractional arm volume (AVol) (cm3); B: Fractional lean arm volume (FLAVol) (cm3); C: Humerus length (cm) and gestational age; NICHD Fetal 3D Study. Estimated 5th, 50th, and 90th percentiles for measurements were estimated from linear mixed models with log-transformed outcomes and cubic splines [Reference 3].
Figure 1. Arm volumes and traditional 2D humerus diaphysis length
Distribution of A: Fractional arm volume (AVol) (cm3); B: Fractional lean arm volume (FLAVol) (cm3); C: Humerus length (cm) and gestational age; NICHD Fetal 3D Study. Estimated 5th, 50th, and 90th percentiles for measurements were estimated from linear mixed models with log-transformed outcomes and cubic splines [Reference 3].
Another area lacking data to guide clinical practice and targeted by our research program is twin pregnancies. Defining abnormal fetal growth in twins is more complicated than for singletons, given that growth restriction can affect one or both twins and can also be defined in terms of discordance. In the NICHD Fetal Growth Studies – Dichorionic Twins, our team developed twin fetal growth percentile charts [Grantz, et al. Am J Obstet Gynecol 2016;215:221]. We found that the fetal growth of twins starts to deviate from that of singletons in the 3rd trimester. This past year, we expanded on these findings to discover that twin growth deceleration begins much earlier in gestation than previously reported using 2D measures. Twin fetuses had less fat and lean tissue than singletons, and they had proportionally less fat throughout pregnancy starting at 15 weeks [Reference 4]. While it has been established that twins are smaller on average than singletons at birth, it was unknown whether growth patterns differed in infancy and childhood. In the first study to compare longitudinal trajectories from birth through 18 years of age, comparing twins and singletons from the same underlying population, we found that twins were smaller in height, weight, and BMI through age 18 [Reference 5]. In addition, unlike growth-restricted singletons, twins had no increased risk of being overweight or obese; the risk for twins was lower throughout childhood and early adolescence, with no difference in risk in late adolescence. Our team has followed up on this work using five years of modern national vital statistics system (NVSS) birth data, to create updated twin birthweight references to be used to identify SGA and LGA among twins, because the previous Alexander reference has errors in gestational age dating. Work is ongoing to develop updated twin birthweight references.
Timing of delivery for pregnancies with complications
Much attention has been focused on preterm delivery, but level–A evidence to guide delivery timing in pregnancies with complications is lacking, an important data gap highlighted by a 2011 joint NICHD workshop. Grantz is co-principal investigator of the Study of Pregnancy and Neonatal Health (SPAN), leading the TIMing of dElivery (TIME) trial to determine the optimal timing of delivery for uncontrolled gestational diabetes mellitus–complicated pregnancies. The study stopped recruiting this past year, and data analysis is in progress.
Labor and delivery management
Our team is also addressing labor and delivery management to prevent medically unnecessary cesarean deliveries, an issue declared as a national priority, given that cesarean delivery is a risk factor for severe maternal morbidity and mortality. The work uses data from the Consortium on Safe Labor Study. Current work targets prolonged second stage of labor because, in prior work, we found that cesareans performed in the second stage of labor are a major contributor to excess cesarean rates [Boyle, et al. Obstet Gynecol 2013;122:33]. Ongoing collaborative work with the Biostatistics and Bioinformatics Branch uses modern statistical approaches not previously used in labor research to build predictive algorithms to apply to the areas of second-stage labor management.
Publications
- Day-specific standard for fetal growth percentiles in the NICHD Fetal Growth Studies-Singletons. Am J Obstet Gynecol 2025 PMID: 41109501;online ahead of print.
- Growth velocity and sonographic estimation of birthweight among low-risk pregnancies. Am J Perinatol 2025 PMID: 40774340;online ahead of print
- Multiethnic growth standards for fetal body composition and organ volumes derived from 3D ultrasonography. Am J Obstet Gynecol 2025 232(3):324
- Fetal body composition in twins and singletons. JAMA Pediatr 2025 179(6):630-638
- Growth trajectories and risk of overweight and obesity in twins and singletons in the UK and USA: analysis of prospective national cohort data. Lancet Child Adolesc Health 2025 9:707-715
Collaborators
- Joseph Biggio, MD, Ochsner Health, Jefferson, LA
- Zhen Chen, PhD, Biostatistics and Bioinformatics Branch, Division of Population Health Research, NICHD
- Celeste Durnwald, MD, Penn Medical, Philadelphia, PA
- Una Grewal, PhD, Division of Population Health Research, NICHD, Bethesda, MD
- Brenna Hughes, MD, Duke University School of Medicine, Durham, NC
- George L. Maxwell, MD, Inova Health System, Fairfax, VA
- Daniel Molina, PharmD, MBA, Technical Resources International, Inc., Bethesda, MD
- Jessica Page, MD, Intermountain Health, Salt Lake City, UT
- Diane Putnick, PhD, Epidemiology Branch, Division of Population Health Research, NICHD, Bethesda, MD
- Robert Silver, MD, University of Utah, Salt Lake City, UT
- Maddy St. Ville, PhD, Biostatistics and Bioinformatics Branch, Division of Population Health Research, NICHD, Bethesda, MD
- Rajeshwari Sundaram, PhD, Biostatistics and Bioinformatics Branch, Division of Population Health Research, NICHD, Bethesda, MD
- Fasil Tekola-Ayele, PhD, Epidemiology Branch, Division of Population Health Research, NICHD, Bethesda, MD
- John Thorp, MD, University of North Carolina, Chapel Hill, NC
- Alan Tita, MD, The University of Alabama at Birmingham, Birmingham, AL
- Edwina Yeung, PhD, Epidemiology Branch, Division of Population Health Research, NICHD, Bethesda, MD
Contact
For more information, email katherine.grantz@nih.gov or visit https://irp.nih.gov/pi/katherine-grantz.