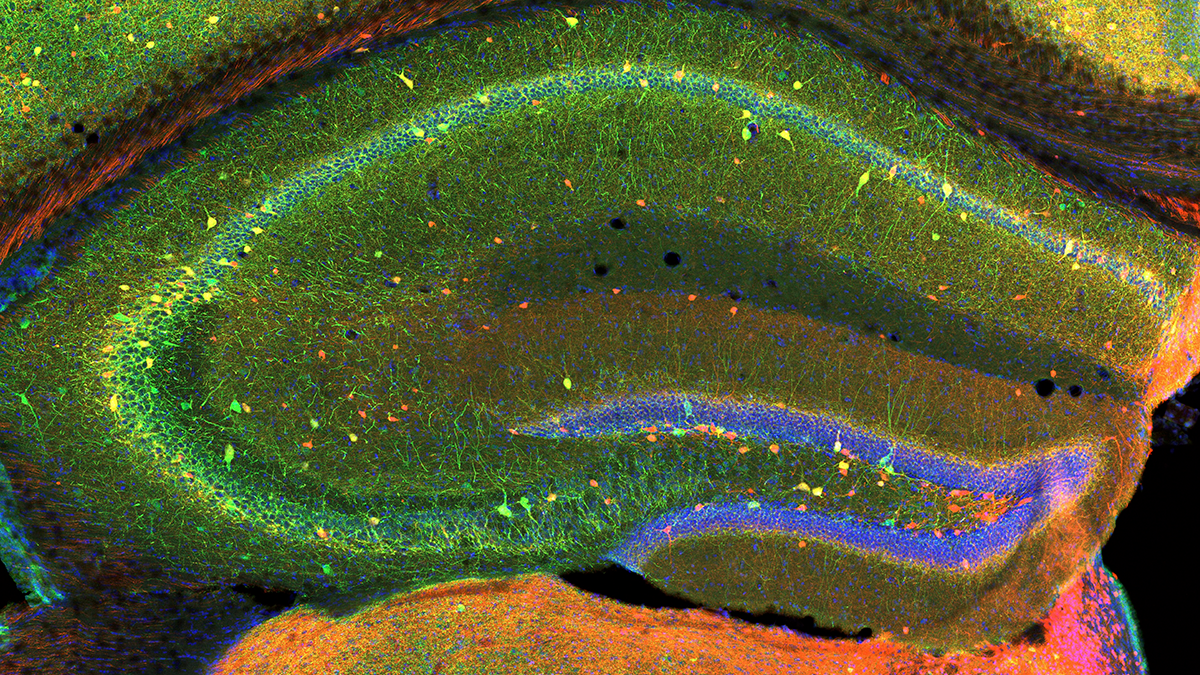
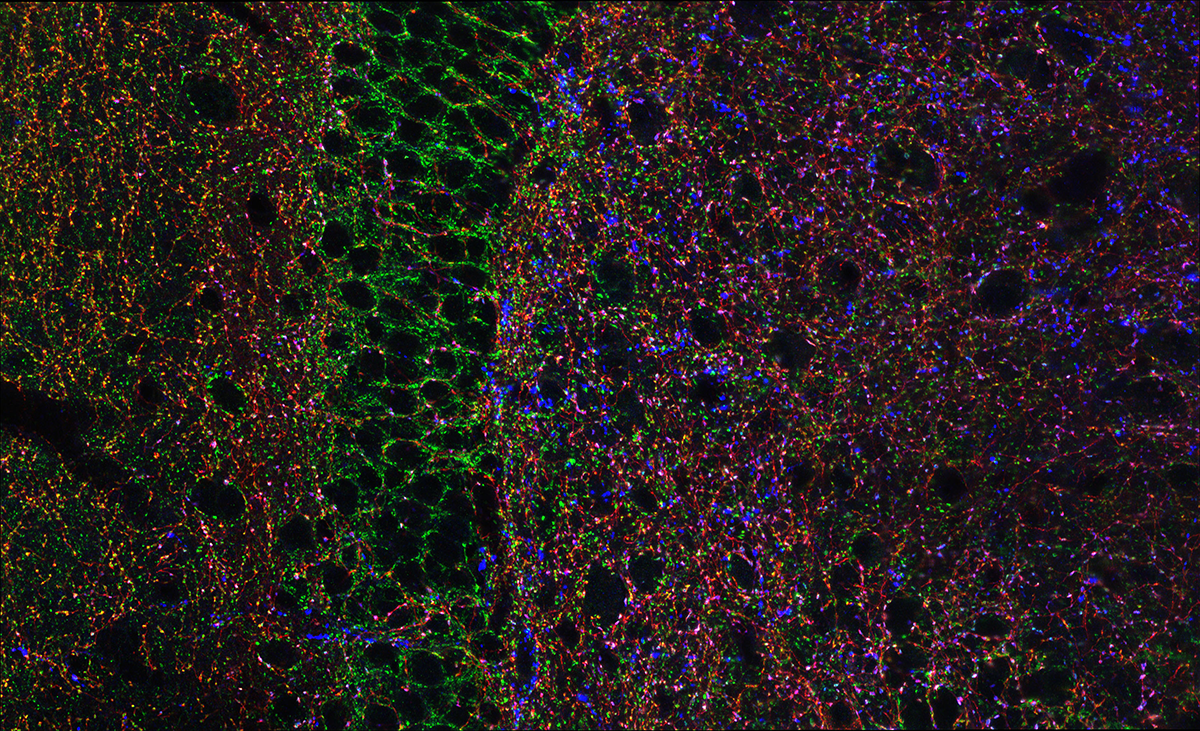
Hippocampal Interneurons and Their Role in the Control of Network Excitability

- Chris J. McBain,
PhD, Head, Section on Cellular and Synaptic Physiology - Ramesh Chittajullu, PhD, Staff Scientist
- Edra London, PhD, Staff Scientist
- Kenneth Pelkey, PhD, Staff Scientist
- Adam Caccavano, PhD, Postdoctoral Fellow
- Lauren Hewitt, PhD, Postdoctoral Fellow
- June Hoan Kim, PhD, Postdoctoral Fellow
- Steven Hunt, BS, Biologist
- Xiaoqing Yuan, MSc, Biologist
- Nadiya McLean, BS, Postbaccalaureate Fellow
- Anna Vlachos, BSc, Postbaccalaureate Fellow
Cortical and hippocampal GABAergic inhibitory interneurons (INs) are ‘tailor-made’ to control cellular and network excitability by providing synaptic and extra-synaptic input to their downstream targets via GABAA and GABAB receptors. The axons of this diverse cell population make mostly local, short-range projections, although some subpopulations project their axons over considerable distances, and release the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) onto a variety of targets. Distinct cohorts of INs regulate sub- and supra-threshold intrinsic conductances, regulate Na+– and Ca2+–dependent action-potential generation, modulate synaptic transmission and plasticity, and pace both local and long-range, large-scale synchronous oscillatory activity. An increasing appreciation of the roles played by INs in several neural-circuit disorders, such as epilepsy, stroke, Alzheimer’s disease, and schizophrenia, has seen this important cell type take center stage in cortical circuit research. With almost 30 years of interest in INs, the main objectives of the lab have been to understand: (1) the developmental trajectories taken by specific cohorts of INs as they populate the nascent hippocampus and cortex; (2) how ionic and synaptic mechanisms regulate the activity of both local-circuit GABAergic INs and principal neurons (PN) at the level of small, well defined networks; and (3) how perturbations in their function alter the cortical network in several neural-circuit disorders. To this end, we use a variety of electrophysiological, imaging, optogenetic, immunohistochemical, biochemical, molecular, and genetic approaches with both wild-type and transgenic animals.
Background and significance
In the hippocampus (HPC), GABAergic local circuit inhibitory INs account for about 10–15% of the total neuronal cell population. Despite being in the minority, this diverse neuronal population serves as a major determinant of all aspects of cortical circuit function and regulation. Within the HPC, the cell bodies of INs are scattered across all major subfields, and the positioning of their somatodendritic arbors allows integration of input from a number of intrinsic and extrinsic afferent inputs. The axons of many IN subtypes largely remain local to the subfield housing their soma and dendrites; however, many form long-range projections that extend beyond their original location to ramify within both cortical and subcortical structures. Their axons target well defined narrow postsynaptic domains (i.e., soma and proximal dendrites) or can provide widespread input to large portions of target cell dendrites. This innervation of different postsynaptic cellular compartments ensures that virtually all domains of their principal neuron targets receive extensive coverage and, importantly, underscores that IN subtypes perform distinct roles in the hippocampal circuit. INs are primarily providers of inhibitory GABAergic synaptic input, a physiological role that utilizes Cl– influx or K+ efflux via cognate GABAA– or GABAB–receptor activation respectively, to transiently hyperpolarize or shunt the cell membrane away from action-potential threshold. These interneurons play major roles in not only the regulation of single-cell excitability, but provide well timed inhibitory input that dictates the temporal window for synaptic excitation and subsequent action-potential initiation, thus shaping the timing of afferent and efferent information flow. In addition, they harness and synchronize both local and distributed cortical circuits to facilitate oscillatory activity across broad frequency domains. Indeed, several developmentally regulated neural circuit disorders, such as epilepsy, schizophrenia, and autism, are likely associated with deficits in the numbers and function of distinct IN cohorts. For all these reasons, INs have recently become the intense focus of investigators drawn from a wide variety of backgrounds.
Our current research has focused on three main aspects of IN function. (1) We continue to study glutamatergic and GABAergic synaptic transmission made onto and from INs and their downstream targets, within the hippocampal and cortical formations. (2) We continue to capitalize on and expand our research using genetic and viral approaches to examine the development of specific cohorts of medial- and caudal-ganglionic eminence–derived INs and their roles in both nascent and mature circuits. (3) As part of a multi-institute consortium, we have expanded our studies to consider evolutionary conservation or diversity of principal-neuron and interneuron function, using tissue derived from a number of species and, in collaboration with NINDS neurosurgery, surgically resected human hippocampal and cortical tissue. This multi-parametric research approach to circuit development and function has been extremely fruitful and is a perfect example of why our research strategy is well suited for the intramural environment. Having the flexibility to pursue this line of research would not have been possible without the support and collaborative nature of the NIH intramural program.
Divergent opioid-mediated suppression of inhibition between hippocampus and neocortex across species and development
Within adult rodent hippocampus (HPC), opioids suppress inhibitory parvalbumin-expressing interneurons (PV–INs), disinhibiting local microcircuits. However, it is unknown whether this disinhibitory motif is conserved across cortical regions, species, or development. We observed that PV–IN–mediated inhibition is robustly suppressed by opioids in the HPC proper but not in the primary neocortex in mice and non-human primates, with spontaneous inhibitory tone in resected human tissue also following a consistent dichotomy. This hippocampal disinhibitory motif is established in early development when PV–INs and opioids regulate early population activity. Morphine pretreatment partially occludes this acute opioid-mediated suppression, with implications for the effects of opioids on hippocampal network activity important for learning and memory. Our findings demonstrate that PV–INs exhibit divergent opioid sensitivity across brain regions, which is remarkably conserved over evolution, and highlight the under-appreciated role of opioids acting through immature PV-INs in shaping hippocampal development.
GluN1/GluN3a excitatory glycine receptor (eGlyR)–mediated tonic currents in hippocampal interneurons control network dynamics throughout development.
The NMDA receptor (N-methyl-D-aspartate receptor, NMDAR) plays a crucial role in learning and memory processes within the brain. As a type of glutamate receptor, the NMDA receptor is involved in the synaptic plasticity that underlies the formation and strengthening of neural connections, so called long-term potentiation, a mechanism believed to be a cellular basis for learning and memory. NMDARs are composed of a combination GluN1, GluN2, GluN3 subunits, and the most common NMDAR types are GluN1/GluN2 diheteromeric receptors. Recently, GluN1/GluN3a excitatory glycine receptor (eGlyR)–mediated currents were reported to tonically influence membrane potential and excitability of neurons in the cortex, medial habenula, and amygdala. These NMDARs are distinct from those expressing GluN2–containing conventional NMDARs, in that they are voltage-independent and gated solely by glycine. Within the HPC, amongst various neuronal types, somatostatin-containing inhibitory interneurons (SST–INs) express remarkably high levels of GRIN3A, a gene encoding GluN3a, throughout development; several subtypes of neurogliaform cells that express the neurotransmitter neuropeptide Y (NPY) cells also express GRIN3A. We determined that the excitability of SST/NPY–IN is strongly influenced by these eGlyRs from early postnatal to adult ages. We found that eGlyR–mediated inward currents in NPY–INs are a major contributor to the GABAergic tone onto pyramidal neurons in the developing HPC, with major consequences for the generation of giant depolarizing potential (GDP). GDPs are rhythmically occurring waves across the neural network that exist between birth to about 10 postnatal days in the HPC and cortex, driving synaptic maturation through activity-dependent plasticity. In the mature HPC, eGlyR–mediated currents in SST–IN eGlyRs also regulate sharp wave ripples (SWRs), network oscillations associated with memory consolidation. Our data are consistent with SST–IN eGlyRs being tonically engaged by endogenous glycine rather than by phasic input from afferents, consistent with prior observations that conventional NMDAR co-agonist binding sites are tonically occupied. Collectively, the data suggest that eGlyRs influence SST/NPY–IN excitability to regulate synchronized network rhythms associated with circuit and memory formation, yielding novel insight into the physiological roles of the notoriously enigmatic GluN3A subunit. In addition, we obtained preliminary data showing that SST–IN excitability is increased by eGlyR activation in the non-human primate HPC, thus influencing SWRs frequency.
Complex opioid-driven modulation of glutamatergic and cholinergic neurotransmission in a GABAergic brain nucleus associated with emotion, reward, and addiction
The medial habenula (mHb)/interpeduncular nucleus (IPN) circuitry contains divergent molecular, neurochemical, and cellular components which, in concert, perform computations to drive emotion, reward, and addiction behaviors. Although housing one of the most prominent mu-opioid receptor (mOR) expression levels in the brain, remarkably little is known about how they impact mHb/IPN circuit function at the granular level. In this study, our systematic functional and pharmacogenetic analyses in mice demonstrated that mOR activation attenuates glutamatergic signaling while producing an opposing potentiation of glutamatergic/cholinergic co-transmission mediated by mHb substance P and cholinergic neurons, respectively. Intriguingly, this latter non-canonical augmentation is developmentally regulated, only emerging during later postnatal stages. In addition, we revealed that specific potassium channels act as a molecular brake on nicotinic receptor signaling in the IPN, with the opioid-mediated potentiation of this arm of neurotransmission being operational only following attenuation of Kv1 (voltage-gated potassium channel) function. Thus, mORs play a complex role in shaping the importance of distinct afferent inputs and transmitter modalities that ultimately influence synaptic recruitment of downstream GABAergic IPN neurons. Together, these observations provide a framework for future investigations aimed at identifying the neural underpinnings of maladaptive behaviors that can emerge when opioids, including potent synthetic analogs such as fentanyl, modulate or hijack this circuitry during the vulnerable stages of adolescence and in adulthood.
A novel and evolutionarily conserved inhibitory circuit selectively regulates dentate gyrus mossy cell function.
The mammalian dentate gyrus (DG) contributes to mnemonic function by parsing similar events and places. The disparate activity patterns of mossy cells and granule cells are believed to enable this function, yet the mechanisms that drive this circuit dynamic remain elusive. We identified a novel inhibitory interneuron subtype, characterized by VGluT3 expression, with overwhelming target selectivity for mossy cells while also revealing that CCK, PV, SST, and VIP interneurons preferentially innervate granule cells. Leveraging pharmacology and novel enhancer viruses, we found that this target-specific inhibitory innervation pattern is evolutionarily conserved in non-human primates and humans. In addition, in vivo chemogenetic manipulation of VGluT3+ interneurons selectively alters the activity and functional properties of mossy cells. These findings establish that mossy cells and granule cells have unique, evolutionarily conserved inhibitory innervation patterns and suggest that selective inhibitory circuits may be necessary to maintain DG circuit dynamics and enable pattern separation across species.
Additional Funding
- Adam Caccavano was partially funded by a Center for Compulsive Disorder Fellowship award.
Publications
- Evolutionary conservation of hippocampal mossy fiber synapse properties. Neuron 2023 111:3802-3818
- Divergent opioid-mediated suppression of inhibition between hippocampus and neocortex across species and development. Neuron 2025 113:1805-1822
- Complex opioid driven modulation of glutamatergic and cholinergic neurotransmission in a GABAergic brain nucleus associated with emotion, reward and addiction. eLife 2025 10.7554:1-20
- An enhancer-AAV toolbox to target and manipulate distinct interneuron subtypes. Neuron 2025 113:1525-1547
Collaborators
- Bruno Averbeck, PhD, Section on Learning and Decision Making, NIMH, Bethesda, MD
- Jordan Dimidschstein, PhD, The Broad Institute, Cambridge, MA
- Gordon Fishell, PhD, The Broad Institute, Cambridge, MA
- Timothy J. Petros, PhD, Unit on Cellular and Molecular Neurodevelopment, NICHD, Bethesda, MD
- Kareem Zaghloul, MD, PhD, Functional and Restorative Neurosurgery Unit, NINDS, Bethesda, MD
Contact
For more information, email mcbainc@mail.nih.gov or visit https://www.nichd.nih.gov/research/atNICHD/Investigators/mcbain.