Spontaneous Preterm Birth and Related Obstetric Syndromes: Integrating Genetic Mechanisms, Cervical Biology, Precision Diagnostics, Clinical Prediction, and Preventive Strategies

- Roberto Romero,
MD, DMedSci, Chief, Pregnancy Research Branch
During the past year, our research program advanced understanding of the biological mechanisms, predictive tools, and preventive strategies underlying major adverse pregnancy outcomes, including spontaneous preterm birth, abnormal labor progression, intra-amniotic infection, and late-onset preeclampsia. Through an integrated portfolio spanning genetics, longitudinal phenotyping, high-dimensional molecular profiling, and clinical data science, we addressed critical gaps across the continuum of obstetric care.
We established cervical remodeling as a genetically regulated biological process that mediates a substantial proportion of inherited risk for spontaneous preterm birth, providing a mechanistic framework linking maternal genetics to parturition timing. Building directly on this biology, we demonstrated robust efficacy and long-term safety of vaginal progesterone for preterm birth prevention across singleton pregnancies and selected high-risk twin gestations, with findings that have directly informed international clinical guidelines.
In parallel, we introduced innovative diagnostic and predictive approaches to improve obstetric decision-making. Real-time nanopore sequencing enabled rapid, species-level diagnosis of intra-amniotic infection and clarified the relationship between microbial burden and intra-amniotic inflammation, advancing precision-medicine approaches for infection-driven preterm labor. Multifactor clinical models substantially improved prediction of labor progress compared with traditional time-based curves, supporting a shift toward data-driven, individualized intra-partum management.
Extending beyond preterm birth, large-scale longitudinal plasma proteomics revealed progressive molecular signatures preceding late-onset preeclampsia and identified multiprotein biomarker panels that outperformed established angiogenic markers in late pregnancy. Together, these studies illustrate a comprehensive, translational research strategy that integrates discovery science with clinical application to improve prediction, prevention, and management of pregnancy complications, with the ultimate goal of reducing maternal and neonatal morbidity worldwide.
Genetic and biological determinants of cervical remodeling underlying spontaneous preterm birth risk
Sonographic cervical length is one of the strongest predictors of spontaneous preterm birth, yet the biological mechanisms governing cervical remodeling across pregnancy have remained incompletely understood. In a large prospective longitudinal cohort of more than 5,000 Black/African American women with serial cervical length measurements and genome-wide genotyping, we investigated the genetic architecture of cervical length dynamics and their relationship with gestational duration.
Using latent growth-curve modeling integrated with genome-wide complex trait analysis, we demonstrated that cervical remodeling across pregnancy is highly heritable, with genetic factors accounting for approximately half of the observed inter-individual variability. Strong genetic correlations between cervical length trajectories and gestational duration indicate that a substantial proportion of genetic risk for spontaneous preterm birth is mediated through biological pathways regulating cervical shortening and effacement.
Genetic loci associated with cervical remodeling were enriched in pathways central to pregnancy maintenance and parturition, including progesterone and estrogen signaling, immune and inflammatory responses, and extracellular matrix organization. These findings establish cervical remodeling as a biologically active, genetically regulated intermediate phenotype linking maternal genetic risk to spontaneous preterm birth, and they provide a framework for integrating genetic information with longitudinal cervical assessment to improve risk prediction and prevention strategies.
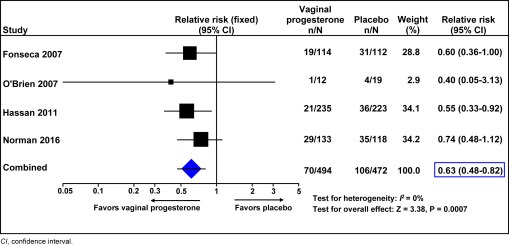
Vaginal progesterone for the prevention of spontaneous preterm birth: robust efficacy across populations and long-term safety
Vaginal progesterone is a cornerstone intervention for the prevention of spontaneous preterm birth in pregnancies at increased risk because of a sonographic short cervix. Through a comprehensive program of research, including updated individual patient data meta-analyses, subgroup analyses, and long-term follow-up studies, we evaluated both the efficacy and safety of vaginal progesterone across diverse clinical populations.
In singleton pregnancies with a mid-trimester transvaginal cervical length less than or equal to 25 mm, vaginal progesterone significantly reduced the risk of early preterm birth across several gestational age thresholds and was associated with reductions in major neonatal morbidity, including respiratory distress syndrome and very low birthweight. Importantly, benefit was observed in women both with and without a prior history of spontaneous preterm birth, resolving a key area of clinical uncertainty and reinforcing the value of universal cervical-length screening followed by targeted treatment.
In selected twin pregnancies with a short cervix, vaginal progesterone also reduced early preterm birth and adverse perinatal outcomes, despite the absence of benefit in unselected twin populations. Long-term follow-up of exposed offspring demonstrated no adverse cognitive, behavioral, or psychopathological effects at six to nine years of age, providing critical reassurance regarding neuro-developmental safety. Together, these studies have directly informed international clinical guidelines and strengthened evidence-based strategies to reduce the global burden of preterm birth.
Rapid diagnosis of intra-amniotic infection using nanopore sequencing technology
Intra-amniotic infection is a major cause of spontaneous preterm labor and preterm, pre-labor rupture of membranes, yet diagnosis is often delayed by the limitations of conventional microbiologic methods. In this prospective cohort study, we evaluated the clinical utility of third-generation nanopore sequencing for rapid, species-level identification of microorganisms in amniotic fluid.
Nanopore sequencing enabled real-time detection of bacterial pathogens, including fastidious and polymicrobial infections, with high diagnostic accuracy and substantially faster turnaround than standard culture-based approaches. The quantitative microbial burden measured by sequencing correlated strongly with the intensity of the intra-amniotic inflammatory response, linking molecular findings to biological severity.
This work demonstrates that real-time sequencing technologies can transform the diagnosis of intra-amniotic infection and provides a foundation for precision approaches to antimicrobial therapy, pregnancy management, and prevention of infection-related preterm birth.
Improving the prediction of labor progress using multifactor clinical models
For decades, assessment of labor progress has relied on time-based labor curves derived from cervical dilation patterns. Although historically influential, these models have limited prospective utility in contemporary clinical practice. In this study, we evaluated whether multifactor clinical models provide more accurate prediction of labor progress.
Using a large longitudinal dataset of more than 8,000 nulliparous women and nearly 48,000 cervical examinations, we compared traditional time-only models with multifactor models incorporating clinically relevant variables such as cervical dilation, effacement, fetal station, and labor interventions. Multifactor models demonstrated substantially greater accuracy and stability, reducing prediction error by nearly 50% compared with time-based approaches.
These findings support reconceptualizing labor as a dynamic, multifactorial process and highlight the potential of data-driven tools to improve intra-partum decision-making, reduce unnecessary interventions, and enhance maternal and neonatal outcomes.
Large-scale plasma proteomics identifies novel biomarkers for late-onset preeclampsia.
Late-onset preeclampsia accounts for the majority of preeclampsia cases worldwide, yet reliable biomarkers for prediction and diagnosis remain limited. Using large-scale longitudinal plasma proteomics, we characterized molecular changes preceding the clinical onset of late-onset preeclampsia across gestation.
High-throughput analysis revealed progressively increasing protein dysregulation with advancing pregnancy, particularly in pathways related to angiogenesis, inflammation, and extracellular matrix remodeling. Multiprotein predictive models constructed from these data outperformed established angiogenic biomarkers in late pregnancy, even before clinical disease became apparent.
These findings demonstrate that late-onset preeclampsia is associated with detectable, progressive molecular alterations in maternal circulation and highlight the potential of proteomics-based approaches to improve risk stratification, diagnosis, and clinical management of hypertensive disorders of pregnancy.
Publications
- Risk factors for spontaneous preterm birth are mediated through changes in cervical length. Commun Med (Lond) 2025 doi: 10.1038/s43856-025-01309-4
- Large-scale proteomics reveals new candidate biomarkers for late-onset preeclampsia. Hypertension 2025 Epub ahead of print
- Vaginal progesterone for the prevention of preterm birth and adverse perinatal outcomes in women with a twin gestation and a short cervix (≤ 25 mm): an updated individual patient data meta-analysis. Ultrasound Obstet Gynecol 2025 66(1):104-107
- A comparison of the accuracy of different labor curves to predict labor progress. Am J Obstet Gynecol. 2025 233(4):e113-e116
- The rapid diagnosis of intraamniotic infection with nanopore sequencing. Am J Obstet Gynecol. 2025 233(3):193.e1-193.e18
Collaborators
- Sergey Bezrukov, DSc, PhD, Section on Molecular Transport, NICHD, Bethesda, MD
- Piya Chaemsaithong, MD, PhD, Mahidol University, Bangkok, Thailand
- Tinnakorn Chaiworapongsa, MD, Wayne State University School of Medicine, Detroit, MI
- Leonid Chernomordik, PhD, Section on Membrane Biology, NICHD, Bethesda, MD
- Agustin Conde-Agudelo, MD, Wayne State University School of Medicine, Detroit, MI
- Amir Gandjbakhche, PhD, Section on Translational Biophotonics, NICHD, Bethesda, MD
- Nardhy Gomez-Lopez, PhD, Washington University School of Medicine, St. Louis, MO
- Narender Kumar, PhD, Center for Biomedical Research, Rockefeller University, Population Council, New York, NY
- Michael W. Kuzniewicz, MD, MPH, University of California School of Medicine, San Francisco, CA
- Larry G. Maxwell, MD, Inova Health System, Falls Church, VA
- Adi L. Tarca, PhD, Wayne State University, Detroit Medical Center, Detroit, MI
- Bruce Tromberg, PhD, Section on Biomedical Optics, NICHD, Bethesda, MD
- Bo Hyun Yoon, MD, PhD, Seoul National University, Seoul, Korea
- Joshua Zimmerberg, MD, PhD, Section on Integrative Biophysics, NICHD, Bethesda, MD
Contact
For more information, email romeror@mail.nih.gov or visit https://irp.nih.gov/pi/roberto-romero.