Thyroid Hormone Regulation of Vertebrate Postembryonic Development

- Yun-Bo Shi,
PhD, Head, Section on Molecular Morphogenesis - Irina Arnaoutova, PhD, Staff Scientist
- Liezhen Fu, PhD, Staff Scientist
- Nga Luu, MS, Biologist
- Suresh Babu Munipalli, PhD, Visiting Fellow
- Alicia Tribondeau, PhD, Visiting Fellow
- Paisley Thomson, PhD, Postdoctoral Fellow
- Emeric Louis, MS, Graduate Student
- Divine Ayichi, BS, Postbaccalaureate Fellow
This laboratory investigates the molecular mechanisms of thyroid hormone (TH) function during postembryonic development, a period around birth in mammals when plasma TH levels peak. The main model is the metamorphosis of pseudo-tetraploid Xenopus laevis and diploid Xenopus tropicalis, two highly related species that offer unique but complementary advantages. The control of this developmental process by TH offers a paradigm to study gene function in postembryonic organ development. During metamorphosis, different organs undergo vastly different changes. Some, like the tail, undergo complete resorption, while others, such as the limbs, are developed de novo. Most larval organs persist through metamorphosis but are dramatically remodeled to function in a frog. For example, the tadpole intestine is a simple tubular structure consisting primarily of a single layer of larval epithelial cells. During metamorphosis, it is transformed into an organ with a multiply folded adult epithelium surrounded by elaborate connective tissue and muscles through specific larval epithelial cell death and de novo development of the adult epithelial stem cells followed by their proliferation and differentiation. The wealth of knowledge from past research and the ability to manipulate amphibian metamorphosis, both in vivo by using genetic approaches or hormone treatment of whole animals, and in vitro in organ cultures, offer an excellent opportunity to: 1) study the developmental function of TH receptors (TRs) and the underlying mechanisms in vivo; and 2) identify and functionally characterize genes that are critical for organogenesis, particularly for the formation of the adult intestinal epithelial stem cells, during postembryonic development in vertebrates. A major recent focus has been to make use of the TALEN and CRISPR/Cas9 technologies to knock out the endogenous genes for functional analyses. In addition, recent improvements in Xenopus tropicalis genome annotation allow us to carry out RNA-Seq and chromatin-immunoprecipitation (ChIP)-Seq analyses at the genome-wide level. They also allows us to adapt single-cell sequencing technology to study how TH induces cell transformations during vertebrate development. Thus, in recent years, we have focused our research on the diploid Xenopus tropicalis. We complement our frog studies by investigating the functions of genes, found to be important for frog intestinal stem-cell development, in developing and adult mouse intestine by making use of the ability to carry out conditional knockout.
Complimentary and additive functions of TRα and TRβ during intestinal remodeling as revealed by ChIP-Seq analysis on wild-type and TR knockout animals
Our earlier studies on TRα and TRβ knockout animals revealed distinct effects of the two TR gene knockouts on limb development, intestinal remodeling, and tail resorption during development. The effects correlate with the relative expression levels of the TRα and TRβ genes during development in these organs, as previously reported by us and others for both Xenopus laevis and Xenopus tropicalis. On the other hand, individual TR knockout does not prevent the eventual completion of metamorphosis and the development of reproductive adult frogs, suggesting that the two TR genes can completement each other. To investigate this further, we used intestinal remodeling during Xenopus tropicalis metamorphosis, which serves as a model for human postembryonic development, to identify TR–bound genes and to determine the relative contribution to target gene binding by TRα and TRβ [References 1, 2]. We first examined the localization of TRα and TRβ mRNA during metamorphosis in Xenopus tropicalis and found that TRα was broadly expressed in the intestinal tissues from premetamorphosis to the end of metamorphosis, while TRβ was expressed at low levels during premetamorphosis but was upregulated at the climax of metamorphosis, when intestinal stem cells are formed and proliferate. Interestingly, both TR genes were co-expressed in different cell types, including stem cells. Chromatin immunoprecipitation (ChIP)-Seq analyses of the intestine from wild-type, TRα– or TRβ–knockout premetamorphic tadpoles treated with or without TH for 18 hours identified many TR–bound genes and revealed the effects of individual TR knockout on the binding of target genes by TR. We found that individual TR knockout reduced both the number of TR–bound genes and the extent of TR binding to target genes, with TRα knockout having a much more dramatic effect than TRβ knockout. On the other hand, the TR–bound genes were largely common among the three genotypes. The findings suggest that both TRα and TRβ contribute to target binding, with TRα having a greater contribution in premetamorphic intestine.
Stage- and TR–dependent gene regulation programs affect the permissive environment during the initial period of Xenopus tropicalis tail regeneration.
Animal regeneration is the natural process of replacing or restoring damaged or missing cells, tissues, organs, and even the entire body to full function. Studies in mammals have revealed that many organs lose regenerative ability soon after birth when TH level is high. This suggests that TH plays an important role in organ regeneration. Intriguingly, plasma TH level peaks during amphibian metamorphosis, which is very similar to postembryonic development in humans. In addition, many organs, such as heart and tail, also lose their regenerative ability during metamorphosis. These make frogs a good model to address how the organs gradually lose their regenerative ability during development and what roles TH may play in this period. Early tail regeneration studies have been carried out mainly in the tetraploid Xenopus laevis (X. laevis), which is a difficult model for gene knockout studies. We have used the highly related but diploid anuran X. tropicalis to investigate the role of TH signaling in tail regeneration with gene knockout approaches. We discovered that X. tropicalis tadpoles could regenerate their tail from premetamorphic stages up to the climax stage 59 and then lose regenerative capacity as tail resorption begins, just like what is observed for X. laevis. To test the hypothesis that TH–induced metamorphic program inhibits tail regeneration, we used TR double knockout (TRDKO) tadpoles lacking both TRα and TRβ, the only two receptor genes in vertebrates, for tail regeneration studies. Our results showed that TRs were not necessary for tail regeneration at any stage in these tadpoles. However, unlike wild-type tadpoles, TRDKO tadpoles retained regenerative capacity at the climax stages 60/61, when wild-type animals lose their tail regeneration capacity. To further investigate this, we carried out RNA-Seq analyses to investigate the gene regulation programs underlying the initiation of tail regeneration, i.e., wound healing and blastema formation [Reference 3]. We discovered that GO (gene ontology) terms related to inflammatory response, metabolic process, cell apoptosis, and epithelial cell migration were highly enriched among commonly regulated genes during wound healing at either stage 56 (when both wild-type and TRDKO animals can regenerate the tail) or stage 61 (when only TRDKO tadpoles can regenerate the tail), consistent with the morphological changes associated with wound healing occurring in both regenerative (wild-type stage 56, TRDKO stage 56, TRDKO stage 61) and non-regenerative (wild-type stage 61) animals. Interestingly, ECM (extracellular matrix)–receptor interaction and cytokine-cytokine receptor interaction were significantly enriched among regulated genes in the three regenerative groups but not in the non-regenerative group at the blastema formation period. In addition, the regulated genes specific to the non-regenerative group were highly enriched with genes involved in cellular senescence, suggesting that TR–mediated, TH–induced gene regulation changed the permissive environment during the initial period of regeneration and affected the patterning/outgrowth period of the regeneration process. Further studies, particularly on the regenerative microenvironment that may depend on ECM–receptor interaction and cytokine-cytokine receptor interaction, should provide important insights on the regulation of regenerative capacity during vertebrate development.
TR is essential for larval epithelial cell death and adult epithelial stem cell development/proliferation during Xenopus tropicalis metamorphosis.
We have shown that knockout of either TRα or TRβ, the only two TR genes, in Xenopus tropicalis, delays but does not prevent intestinal remodeling, including stem cell formation and/or proliferation. On the other hand, TR double knockout (TRDKO) Xenopus tropicalis tadpoles fail to undergo either larval epithelial cell death or adult epithelial stem cell development and proliferation by stage 61, the climax stage when TRDKO animals die. Interestingly, adult epithelial morphogenesis occurs prematurely in TRDKO tadpoles. To investigate how TR regulates cell fate and tissue morphogenesis, we are now using scRNA-Seq and snRNA-Seq to determine how TH regulates the changes within different types of cells in the intestine during metamorphosis [Reference 4].
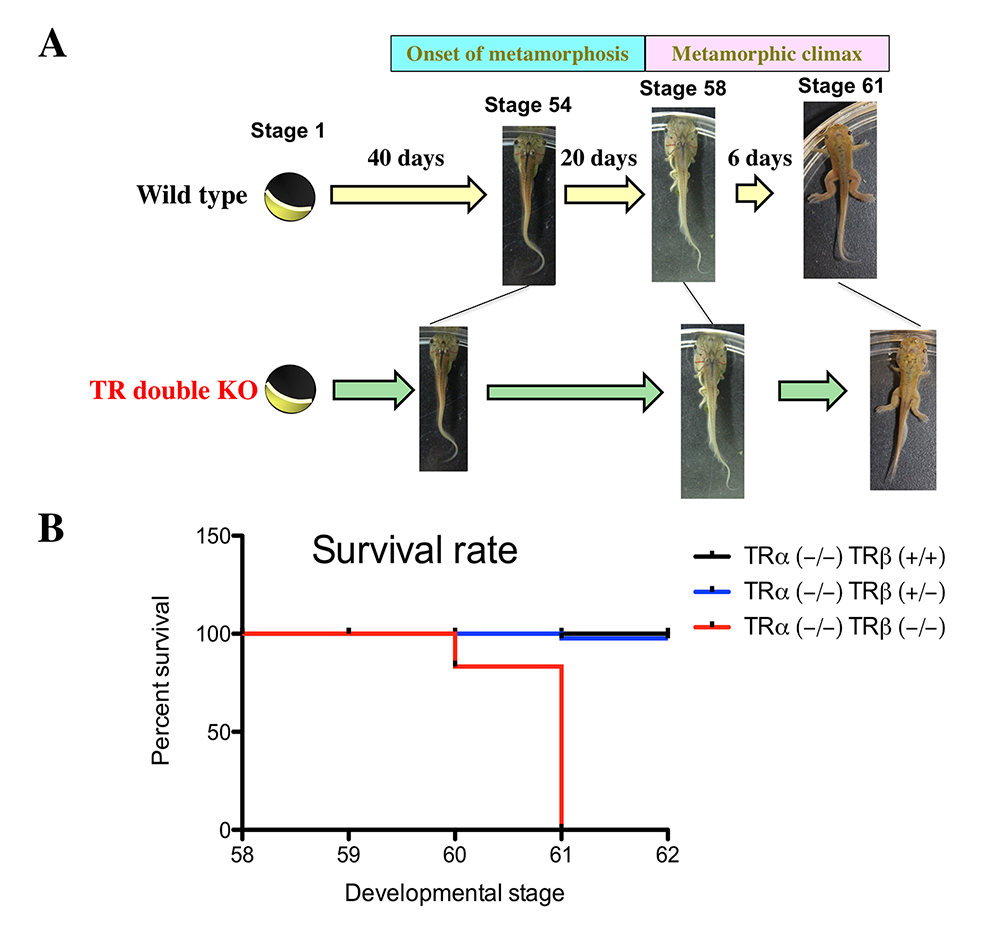
Figure 1. Effects of TR double KO on developmental rate: TR double KO leads to premature initiation of metamorphosis but slows metamorphic progress and causes lethality at the metamorphic climax.
A. TR double KO animals take a shorter time to reach the onset of metamorphosis (stage 54), indicating accelerated pre-metamorphic development. Once metamorphosis begins, the KO animals take longer to reach the beginning of metamorphic climax (stage 58) and also develop more slowly during the climax stages, between stages 58 and 61. The length of each stage indicates the relative time needed for development between two adjacent stages.
B. Tadpoles without any TR die during the climax of metamorphosis. The tadpoles of mixed genotypes at stage 58 were able to develop to stage 62 and were genotyped at stage 62 or when they died during this developmental period. The survival rate for each of the three genotypes, trα–/–trβ+/+, trα–/–trβ+/–, and trα–/–trβ–/–, was thus obtained and plotted. Note that no double KO tadpoles developed to stage 62 but that a single copy of trβ+/– was sufficient for the animal to complete metamorphosis and develop into a reproductive adult.
Figure 1. Effects of TR double KO on developmental rate: TR double KO leads to premature initiation of metamorphosis but slows metamorphic progress and causes lethality at the metamorphic climax.
A. TR double KO animals take a shorter time to reach the onset of metamorphosis (stage 54), indicating accelerated pre-metamorphic development. Once metamorphosis begins, the KO animals take longer to reach the beginning of metamorphic climax (stage 58) and also develop more slowly during the climax stages, between stages 58 and 61. The length of each stage indicates the relative time needed for development between two adjacent stages.
B. Tadpoles without any TR die during the climax of metamorphosis. The tadpoles of mixed genotypes at stage 58 were able to develop to stage 62 and were genotyped at stage 62 or when they died during this developmental period. The survival rate for each of the three genotypes, trα–/–trβ+/+, trα–/–trβ+/–, and trα–/–trβ–/–, was thus obtained and plotted. Note that no double KO tadpoles developed to stage 62 but that a single copy of trβ+/– was sufficient for the animal to complete metamorphosis and develop into a reproductive adult.
TH regulation of cell transformations during liver metamorphosis in Xenopus tropicalis
Many mammalian organs and tissues, including erythrocytes, mature into their adult forms during postembryonic development when plasma TH level peaks, resembling amphibian metamorphosis. TR mutations/deletions can cause hematopoietic dysfunction, suggesting that TH plays a role in erythropoiesis during development. Our analyses of TR double knockout (TRDKO) Xenopus tropicalis revealed that TRDKO tadpoles exhibited characteristics similar to human iron deficiency anemia. As the liver is the hematopoietic organ, our finding suggests a defect in liver development in TRDKO tadpoles. Indeed, we found that TRDKO tadpoles had liver developmental defects during metamorphosis, including reduced cell proliferation and failure to undergo hepatocyte hypertrophy or activate the expression of urea cycle genes. To investigate how TR regulates the transformations of different cell types in the liver during metamorphosis, we are carrying scRNA-Seq and snRNA-Seq analyses of liver during natural and T3–induced metamorphosis of wild-type and TRDKO tadpoles.
Protein arginine methyltransferase 1 regulates mouse intestinal epithelial homeostasis in mouse.
Earlier studies suggested that the formation of adult mouse intestinal stem cells takes place during the first few weeks after birth, the postembryonic period when plasma TH levels are high. Furthermore, deficiency in TH signaling leads to defects in adult mouse intestine, including reduced cell proliferation in the intestinal crypts, where stem cells reside. Thus, mouse intestinal maturation resembles intestinal remodeling during frog metamorphosis, both in morphological development and in the likely role of TH in the process. Our previous studies showed that protein arginine methyltransferase 1 (PRMT1), a TR coactivator, is important for adult intestinal stem cell development and/or proliferation during Xenopus metamorphosis. We further showed that PRMT1 is also highly expressed during intestinal maturation in the mouse, resembling its upregulation during the intestinal metamorphosis of the frog. Importantly, we analyzed the expression of PRMT1 by immunohistochemistry and studied the effect of tissue-specific knockout of PRMT1 in the intestinal epithelium. We showed that PRMT1 was highly expressed in the proliferating transit amplifying cells and crypt-base stem cells. The specific knockout of PRMT1 in the intestinal epithelium resulted in, surprisingly, more elongated adult intestinal crypts with increased cell proliferation in the adult mice. In addition, Goblet cells were increased and Paneth cells were reduced in the crypt. Furthermore, using a tamoxifen-inducible intestinal epithelium–specific PRMT1 knockout mouse model, we showed that the epigenetic enzyme PRMT1 is important for the maintenance of intestinal epithelial architecture and homeostasis [Reference 5]. We are currently investigating whether PRMT1 also influences intestinal development, particularly via T3 signaling, by using morphological, molecular, and snRNA-Seq analyses.
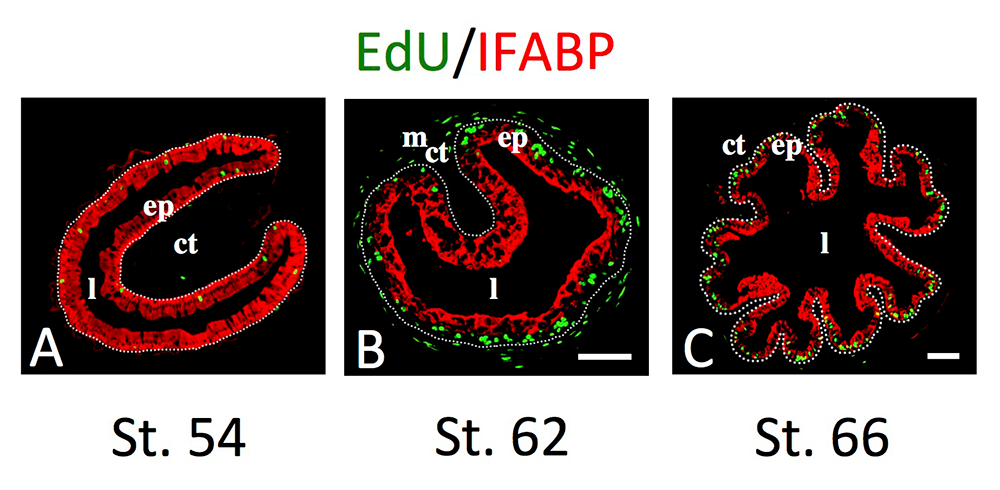
Figure 2. Intestinal metamorphosis involves the formation of clusters of proliferating, undifferentiated epithelial cells at the climax.
Tadpoles at pre-metamorphic stage 54 (A), climax, stage 62 (B), and the end of metamorphosis, stage 66 (C) were injected with 5-ethynyl-2′-deoxyuridine (EdU) one hour before sacrifice. Cross-sections of the intestine from the resulting tadpoles were double-stained by EdU labeling of newly synthesized DNA and by immunohistochemistry of IFABP (intestinal fatty acid–binding protein), a marker for differentiated epithelial cells. The dotted lines depict the epithelium-mesenchyme boundary. Note that there are few EdU–labeled proliferating cells in the epithelium and that they express IFABP at pre-metamorphosis (A) and increase in the form of clustered cells (proliferating adult stem cells), which lack IFABP at the climax of metamorphosis (B). At the end of metamorphosis, EdU–labeled proliferating cells are localized mainly in the troughs of the epithelial folds, where IFABP expression is low (C). ep, epithelium; ct, connective tissue; m, muscles; l, lumen.
Figure 2. Intestinal metamorphosis involves the formation of clusters of proliferating, undifferentiated epithelial cells at the climax.
Tadpoles at pre-metamorphic stage 54 (A), climax, stage 62 (B), and the end of metamorphosis, stage 66 (C) were injected with 5-ethynyl-2′-deoxyuridine (EdU) one hour before sacrifice. Cross-sections of the intestine from the resulting tadpoles were double-stained by EdU labeling of newly synthesized DNA and by immunohistochemistry of IFABP (intestinal fatty acid–binding protein), a marker for differentiated epithelial cells. The dotted lines depict the epithelium-mesenchyme boundary. Note that there are few EdU–labeled proliferating cells in the epithelium and that they express IFABP at pre-metamorphosis (A) and increase in the form of clustered cells (proliferating adult stem cells), which lack IFABP at the climax of metamorphosis (B). At the end of metamorphosis, EdU–labeled proliferating cells are localized mainly in the troughs of the epithelial folds, where IFABP expression is low (C). ep, epithelium; ct, connective tissue; m, muscles; l, lumen.
Publications
- Thyroid hormone receptor subtype-specific function in controlling organ-specific developmental timing and rate during Xenopus development. Front Endocrinol 2025 16:1614439
- Functions and mechanism of thyroid hormone receptor action during amphibian development. Endocrinology 2024 165:bqae137
- Thyroid hormone receptor- and stage-dependent transcriptome changes affect the initial period of Xenopus tropicalis tail regeneration. BMC Genomics 2024 25:1260
- Complementary and additive functions of TRα and TRβ during intestinal remodeling as revealed by ChIP-Seq analysis on wild type and TR knockout animals. Gen Comp Endocrinol 2025 360:114645
- Protein arginine methyltransferase 1 is required for the maintenance of adult small intestinal and colonic epithelial cell homeostasis. Int J Biol Sci 2024 20:554-568
Collaborators
- Ryan Dale, MS, PhD, Bioinformatics and Scientific Programming Core, NICHD, Bethesda, MD
- Caroline Esnault, PhD, Bioinformatics and Scientific Programming Core, NICHD, Bethesda, MD
- James Iben, PhD, Molecular Genomics Core, NICHD, Bethesda, MD
- Tianwei Li, PhD, Molecular Genomics Core, NICHD, Bethesda, MD
- Vivek Mahadevan, PhD, Molecular Genomics Core, NICHD, Bethesda
- Fabio Rueda Faucz, PhD, Molecular Genomics Core, NICHD, Bethesda, MD
- Laurent Sachs, PhD, Centre National de la Recherche Scientifique (CNRS), Paris, France
- Bingyin Shi, MD, Xi’an Jiaotong University School of Medicine, Xi'an, China
- Guihong Sun, PhD, Wuhan University School of Medicine, Wuhan, China
- Chuan Wu, MD, PhD, Experimental Immunology Branch, NCI, Bethesda, MD
- Henry Zhang, PhD, Bioinformatics and Scientific Programming Core, NICHD, Bethesda, MD
Contact
For more information, email shi@helix.nih.gov or visit https://www.nichd.nih.gov/research/atNICHD/Investigators/shi.