Chemosensory Coding and Decoding by Neuron Ensembles

- Mark Stopfer,
PhD, Head, Section on Sensory Coding and Neural Ensembles - Zane Aldworth, PhD, Staff Scientist
- Bo-Mi Song, PhD, Staff Scientist
- Yuzhong Cheng, PhD, Senior Research Assistant
- Kui Sun, MD, Research Assistant
- Matthew Brown, PhD, Postdoctoral Fellow
- Leah Pappalardo, BS, Graduate Student
- Kelechi Akalaonu, BS, Postbaccalaureate Fellow
All animals need to know what is going on in the world around them. Brain mechanisms have thus evolved to gather and organize sensory information to build transient and sometimes enduring internal representations of the environment.
Using relatively simple animals and focusing primarily on olfaction and gustation, we combine electrophysiological, anatomical, behavioral, computational, optogenetic, and other techniques to examine the ways in which intact neural circuits, driven by sensory stimuli, process information. Our work reveals basic mechanisms by which sensory information is transformed, stabilized, and compared, as it makes its way through the nervous system.
We use three species of insects, each with specific and interlocking experimental advantages, as our experimental preparations: locusts, moths, and fruit flies. Compared with vertebrates, the insect nervous system contains relatively few neurons, most of which are readily accessible for electrophysiological study. Essentially intact insect preparations perform robustly following surgical manipulations, and insects can be trained to provide behavioral answers to questions about their perceptions and memories. Ongoing advances in genetics permit us to target specific neurons for optogenetic or electrophysiological recording or manipulations of activity. Furthermore, the relatively small neural networks of insects are ideal for tightly constrained computational models that test and explicate fundamental circuit properties.
Development of olfactory behaviors
The sense of smell is essential for survival throughout the lifespan of most animals, from the moment of birth on. Studying the olfactory system as an animal develops can reveal fundamental aspects of information processing by networks of neurons as they grow and interact with the environment. The study of relatively simple animals such as locusts has proved to be a useful strategy for answering basic questions about olfaction.
Many animals, including humans, display innate preferences for some odors, but the neural mechanisms underlying these preferences are poorly understood. To investigate the encoding of innate preferences, it is necessary to establish a model system that allows both clear behavioral demonstrations of olfactory preferences and tractable analyses of neural mechanisms. Thus, using locusts that had just hatched, we designed and used an open-field arena to provide only olfactory cues to guide navigation choices, and we used our own Argos tracking software to quantify the movements of naive hatchlings. We found that newly hatched locusts navigated toward, and spent more time near, the odor released by a typical locust food, wheat grass, than near humidified air. Notably, in similar tests, we found that hatchlings avoided moderate concentrations of major individual components of the wheat grass odor, 1-hexanol and hexanal diluted in mineral oil, compared with control presentations of unscented mineral oil. Hatchlings were neither attracted to nor repelled by a lower concentration of 1-hexanol but were moderately attracted to a low concentration of hexanal.
Our behavioral results provide the first unambiguous demonstration that fresh-from-the-egg locust hatchlings have a strong, innate preference for the odor of wheat grass (a blend of many components), but that the valence of the blend’s individual components may be different and may change depending on the concentration. Our results provide an essential entry point for an analysis of neural mechanisms underlying innate sensory preferences.
As locusts grow from hatchling to adulthood, they acquire experience with the olfactory environment, and their body size increases tenfold while their brain size almost triples. Understanding how these changes affect the processing of olfactory information requires a rigorous investigation of the structure and function of developing locusts.
We therefore conducted an analysis of the structure and function of the first stages in the olfactory pathway in newly hatched and adult locusts by making electrophysiological recordings from their neurons to characterize their responses to odors and by filling them with dye to visualize their morphologies. Our quantitative comparisons indicate that, although smaller in size, hatchling olfactory neurons are functionally and structurally similar to those of adults. With electro-antennograms elicited by a broad panel of odors, we determined that the tuning of the olfactory-receptor neuron population is similar in hatchlings and adults. Furthermore, with patch-clamp recordings, we found that local and projection neurons of the antennal lobe respond to odors with quantitatively similar complex, synchronous, oscillatory activity patterns in hatchlings and adults.
Notably, we found that odor-elicited neural oscillations in hatchlings gradually increase in frequency as the locusts grow. Based on our earlier computational modeling results, we hypothesized that this frequency change could be explained by increased odor-elicited excitatory drive, which accrues as new olfactory receptor neurons are added to growing antennae. To test this hypothesis, we successively removed segments containing olfactory receptor neurons from the adult antenna while recording local field potentials from the mushroom bodies. Consistent with our hypothesis, removing antennal segments significantly reduced the frequency of odor-elicited neural oscillations only on the side of the brain ipsilateral to the clipped antenna.
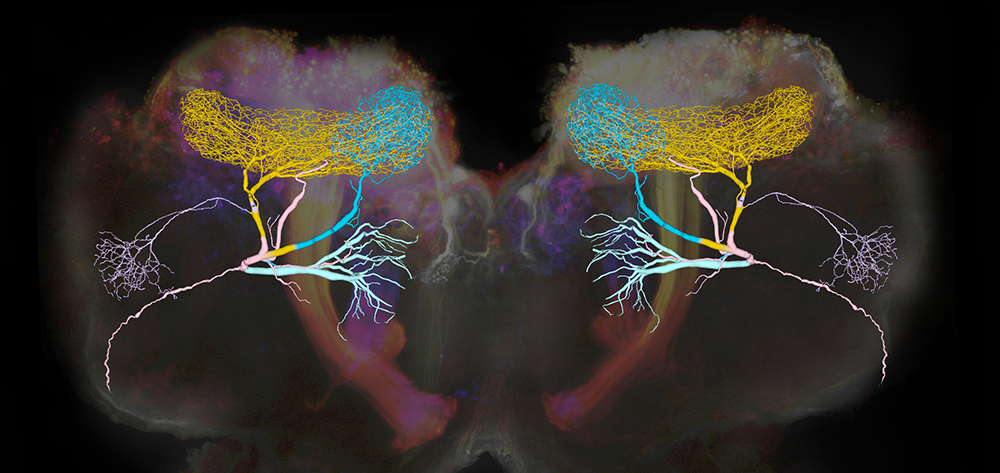
Figure 1. Giant GABAergic neurons regulate olfactory responses in the locust brain.
The composite image shows the structure of a compartmental computational model of the giant GABAergic neurons (GGNs) superimposed on dextran-dyed mushroom bodies in the locust brain. Different branches of GGN are shown in different colors. GGNs, only one on each side of the brain, regulate the firing of tens of thousands of olfactory neurons through feedback inhibition.
Figure 1. Giant GABAergic neurons regulate olfactory responses in the locust brain.
The composite image shows the structure of a compartmental computational model of the giant GABAergic neurons (GGNs) superimposed on dextran-dyed mushroom bodies in the locust brain. Different branches of GGN are shown in different colors. GGNs, only one on each side of the brain, regulate the firing of tens of thousands of olfactory neurons through feedback inhibition.
Development of olfactory system structure and function
An important question in developmental neuroscience is how sensory systems change as animals grow and interact with the environment. Exploring sensory systems as they develop can reveal how networks of neurons process information as the neurons themselves grow and the needs of the animal change. We compared the structure and function of peripheral parts of the olfactory pathway in newly hatched and adult locusts. We found that populations of olfactory sensory neurons (OSNs) in hatchlings and adults responded with similar tunings to a panel of odors. The OSNs send olfactory information to the antennal lobes (ALs). The morphologies of local neurons (LNs) and projection neurons (PNs) in the ALs were very similar in both age groups; though they were smaller in hatchlings, they were proportional to overall brain size. The odor-evoked responses of LNs and PNs were also very similar in both age groups, characterized by complex patterns of activity including oscillatory synchronization. Notably, in hatchlings, spontaneous and odor-evoked firing rates of PNs were lower, and LFP (local field potential) oscillations were lower in frequency, than in the adult. Like many young animals, hatchlings have fewer OSNs than adults. Removing antennal segments from adults also reduced LFP oscillation frequency. Thus, consistent with our earlier computational models, the developmental increase in frequency is attributable to increasing intensity of input to the oscillation circuitry. Overall, our results show that locusts hatch with a fully formed olfactory system that structurally and functionally matches that of the adult, despite its small size and lack of prior experience with olfactory stimuli.
The brain matches neural information coding formats to different sensory tasks.
All animals must detect information from their environment and convert it into signals that can be processed by the brain. The sense of smell provides a powerful model for studying how sensory information is encoded, and insects offer especially useful and relatively simple systems for this kind of research. In this study, we took advantage of a key feature of insect olfaction: insects have two main smell organs, one on the antennae and another on the mouthparts, which appear to have different roles.
Little is known about the olfactory pathway that begins in the mouthparts, including how it encodes information and what functions it supports. Using a comprehensive anatomical approach, we traced the neural pathways from odor-sensing neurons in the mouthparts through multiple layers of the brain. With an electrophysiological approach, we also measured how these neurons respond to different types of odorants. We then compared these results with those from the much better understood antennal olfactory pathway.
Our analysis revealed that the two pathways serve different purposes and process odor information in distinct ways. The antennal pathway functions as a general-purpose odor detection system and uses a coding strategy well suited to distinguishing, comparing, and remembering a wide range of smells. In contrast, the mouthpart pathway is simpler and more specialized. It responds mainly to odors associated with food and appears to work as an adjunct to the taste system, using a much less complex coding format. Together, these findings show that a single animal can use different “languages” to process the same type of sensory information. Rather than relying on a single neural coding strategy, the brain employs different coding approaches tailored to the specific demands of different tasks.
A peripheral feedforward neural circuit amplifies taste signals to enhance behavioral reliability.
A fundamental goal of neuroscience is to understand how sensory systems transform signals from the outside world into neural activity that reliably guides behavior. In this project, we describe a previously unknown neural circuit at the sensory periphery that integrates and amplifies taste signals, making taste-driven behaviors more consistent.
Using anatomical mapping and electrical recordings, we found five taste-sensing neurons in the fruit fly Drosophila melanogaster that form monosynaptic connections to one another. These neurons respond to similar taste cues, and four of them form directional, selective, and functional excitatory connections that work together to boost incoming taste information. This arrangement creates a feedforward amplification circuit that increases the reliability of taste signals.
We further discovered that communication between these taste neurons depends largely on a specific receptor, the nicotinic acetylcholine receptor α5 subunit. Removing this receptor genetically did not change how long flies fed on average, but it made their feeding behavior significantly more variable from trial to trial. Restoring the receptor reversed this effect, returning behavior to a more consistent pattern.
Taken together, our results uncover a previously unknown neural circuit made up of identifiable neurons that amplifies taste information at the earliest stage of processing. This mechanism helps ensure reliable behavior, highlighting how robustness can be built directly into peripheral sensory systems.
Publications
- Olfactory system structure and function in newly hatched and adult locusts. Sci Rep 2024 14:2608
- Olfactory receptor neurons generate multiple response motifs, increasing coding space dimensionality. eLife 2023 12:e79152
- Innate attraction and aversion to odors in locusts. PLoS ONE 2023 18(7):e0284641
Collaborators
- Timothy Petros, PhD, Unit on Cellular and Molecular Neurodevelopment, NICHD, Bethesda, MD
Contact
For more information, email stopferm@mail.nih.gov or visit https://www.nichd.nih.gov/research/atNICHD/Investigators/stopfer.