You are here: Home > Section on Growth and Obesity
Physiology, Psychology, and Genetics of Obesity

- Jack A. Yanovski, MD, PhD, Head, Section on Growth and Obesity
- Joan C. Han, MD, Assistant Clinical Investigator
- Asem Ali MD, Clinical Fellow, Endocrine Training Program
- Melissa K. Crocker, MBA, MD, Clinical Fellow, Endocrine Training Program
- Lauren B. Shomaker, PhD, Special Volunteer
- Roya Sherafat, MD, PhD, Special Volunteer
- Diane C. Adler-Wailes, MS, Biologist
- Sheila M. Brady, RN, FNP, Nurse Practitioner
- Shannon Fuhr, BA, Postbaccalaureate Fellow
- Caroline A. Roza, BS, Postbaccalaureate Fellow
- Yasmmyn Salinas, BS, Postbaccalaureate Fellow
- Mahsa Sorouri, BS, Postbaccalaureate Fellow
- Babette Stern, BS, Postbaccalaureate Fellow
- Jaclyn Zocca, BA, Postbaccalaureate Fellow
The prevalence of overweight and obesity in children and adults has tripled during the past 40 years. The alarming rise in body weight has likely occurred because the current environment affords easy access to calorie-dense foods and requires less voluntary energy expenditure. However, this environment leads to obesity only in those individuals whose body weight–regulatory systems are not able to control body adiposity with sufficient precision in our high-calorie/low-activity environment, suggesting that there are subgroups in the U.S. have a uniquely high susceptibility to weight gain under prevailing environmental conditions. The primary goal of the Section on Growth and Obesity is to elucidate the genetic underpinnings of the metabolic and behavioral endophenotypes that contribute to the development of obesity in children. Drawing on our unique longitudinal cohort of children at risk for adult obesity who have undergone intensive metabolic and behavioral phenotyping, we examine genetic and phenotypic factors predictive of progression to adult obesity in children in the "pre-obese" state, allowing characterization of phenotypes unconfounded by the impact of obesity itself. Once identified as linked to obesity, genetic variants that impair gene function undergo intensive study. Such approaches are expected to improve our ability to predict which children are at greatest risk for obesity and its comorbid conditions and to lead to more targeted, etiology-based prevention and treatment strategies for pediatric obesity.
Studies of genetic factors important for childhood body weight regulation
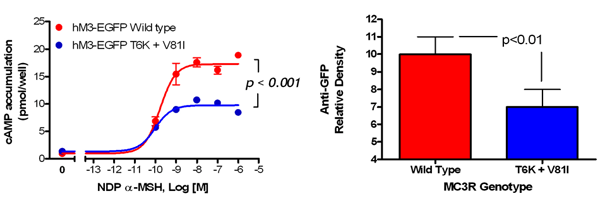
Figure 1
Studies of a human MC3R variant containing two naturally occurring polymorphisms—a variant associated with pediatric-onset obesity—found that the variant was partially inactive, with decreased signal transduction (left panel), likely due to reduced protein expression (right panel).
To identify gene variants affecting body composition, we have been examining polymorphisms in genes involved in the leptin signaling pathway. Genes include FTO and those encoding proopiomelanocortin (POMC), the melanocortin 3 receptor (MC3R), brain-derived neurotrophic factor (BDNF), and histaminergic receptors 1 and 3. We are currently studying a variant MC3R that is associated with adiposity in children and appears to have functional significance for MC3R signal transduction. Children who were homozygous variant for both Thr6Lys and Val81Ile polymorphisms had significantly greater BMI-SD scores, fat mass, and body circumference measurements and higher plasma levels of insulin and leptin than unaffected or heterozygous children. In vitro studies subsequently found that signal transduction and protein expression were significantly lower for the double mutant MC3R (Figure 1). Our ongoing studies attempt to understand the mechanisms by which these sequence alterations may affect body weight. We found that children homozygous variant for both Thr6Lys and Val81Ile polymorphisms showed no deficits in energy expenditure but demonstrated hyperphagia in laboratory meal studies (Figure 2). The results were specific to function-altering mutations and not associated with other common polymorphisms we identified in the MC3R. Transgenic "knock-in" mice expressing the human wild-type and human double-mutant MC3R have been developed in collaboration with Heiner Westphal for study over the next two years.

Click image to enlarge.
Figure 2
Energy intake is studied by using free-access buffet meals of palatable foods. Children homozygous for two polymorphisms in the MC3R consumed more at the buffet than heterozygotes or those with wild-type MC3R.
We also recently investigated the BDNF-TrkB pathway in relation to body mass in children. We measured serum BDNF in 328 children, age 3–19 years, many exhibiting extreme obesity. BDNF was significantly lower in obese children; in multiple regression analyses with log-BDNF as the dependent variable, BMI, BMI-Z, and body fat were all negatively associated with BDNF. The data suggest that some obese individuals with low serum BDNF for age may have mutations that alter BDNF function.

Click image to enlarge.
Figure 3
Patients with WAGR syndrome who have haploinsufficiency for brain-derived neurotrophic factor (BDNF) had a higher BMI standard deviation score (BMI Z-Score) than children and adults with WAGR syndrome who retained two copies of BDNF. Deletions that extended into exon 1 of BDNF were associated with 100% risk of childhood-onset obesity.
Another initiative assessed the role of BDNF haploinsufficiency as a cause of obesity in patients with syndromes attributable to deletions in the vicinity of 11p14.1, where the human BDNF gene is located. In 33 subjects with the WAGR (Wilms tumor, aniridia, genitourinary, and renal abnormalities) syndrome who had heterozygous 11p deletions, ranging in size from 1.0–26.5 Mb, 19 had regions of deletion that involved the BDNF gene (BDNF+/−). Compared with those with intact BDNF (BDNF+/+), BDNF+/− had significantly greater body mass during childhood, starting at age 2 (Figure 3). 100% of BDNF+/− were obese by age 10 versus only 20% of BDNF+/+. Analysis of the telomeric deletion boundaries of patients indicated the presence of a critical region for pediatric-onset obesity within 80 kb of BDNF exon (Figure 3). Each subject who maintained normal weight during childhood had a deletion that did not involve BDNF. These findings suggest that BDNF haploinsufficiency in patients with WAGR syndrome leads to pediatric-onset obesity. A clinical protocol admitting patients with WAGR syndrome and other 11p deletion syndromes for full characterization of their energy intake and expenditure is now under way.
We have also been investigating conditions such as Bardet Biedl and Alström syndromes that, based on mouse model data, may impair leptin receptor function. In patients with Bardet Biedl syndrome, we found hyperleptinemia consistent with leptin receptor resistance, suggesting that defect-specific therapy may be possible.
Physiology, metabolism, and psychology of childhood body weight regulation
Our studies are directed at understanding the physiological, psychological, and metabolic factors that place children at risk for undue weight gain. As part of these studies, we examined how best to measure eating-related psychopathology, insulin sensitivity, and changes in body composition, energy intake, and energy expenditure in children. We also studied the short- and long-term stability of the components of metabolic syndrome. Longitudinal studies demonstrated that leptin is an important predictor of weight gain in children. Recent investigations also documented how hyperinsulinemia is related to energy intake in non-diabetic, obese children who selected lunch from a 9,835kcal buffet eaten ad libitum after an overnight fast. Energy intake was positively associated with the fasting homeostasis model assessment for insulin resistance index (HOMA-IR), fasting insulin/glucose ratio, first-phase insulin, and first-phase C-peptide during a hyperglycemic clamp; energy intake was negatively associated with clamp-derived insulin sensitivity (SIclamp). Each 10% decrease in SIclamp predicted 27 kcal greater energy intake. We also examined the relationship between depressive symptomatology and insulin resistance in children and adolescents, finding strong associations both cross-sectionally and prospectively between depressive symptoms and insulin resistance, independent of body weight. These associations not only suggest mechanisms whereby insulin resistance may contribute to excessive weight gain in children but have also informed some of our treatment approaches to pediatric obesity (described below).
Our evaluations of binge-eating behaviors in children suggest that such behaviors are also associated with adiposity in children. We found that binge-eating and dieting behaviors may predict future weight gain in children at risk for obesity; children reporting binge-eating behaviors such as loss of control over eating gained an additional mean 2.4 kg per year compared with non–binge-eating children. Our data also suggest that children endorsing binge eating consume more energy during meals. Actual intake during buffet meals averaged 400 kcal more in children exhibiting binge eating, but such children reported shorter-lived satiety than children who did not engage in binge-eating episodes. The ability to consume large quantities of palatable foods, especially when coupled with decreased subsequent satiety, may play a role in the greater weight gain in binge-eating children. The data also suggest that interventions targeting disordered eating behaviors may be useful in preventing excessive fat gain in children prone to obesity. Given that binge eating appears to be an African American heritable trait, we initiated studies to investigate potential genetic factors linked to loss of control over eating (3). We genotyped 229 youth 6–19 years for FTO SNP rs9939609, performed body-composition measurements, and interviewed them to determine the presence or absence of loss-of-control (LOC) eating. Subjects with at least one A allele (67.7%) had significantly greater BMI, BMI Z-scores, and fat mass. Of the AA/AT subjects, 34.7% reported LOC eating compared with 18.2% of the TT (wild-type) subjects. AA/AT subjects consumed a significantly greater percentage of energy from fat than did the TT subjects.
To determine the most important factors for the development of obesity complications in youth, we study, in two ongoing protocols, normal-weight children and adolescents, already obese children, and the non-obese children of obese parents. We examine body composition, metabolic rate, insulin sensitivity, glucose disposal, energy intake at buffet meals, and genetic factors believed to regulate metabolic rate and body composition. We also investigate psychological and behavioral factors, such as propensity to engage in binge-eating behavior. As we follow children into adulthood, we are studying actual food consumption of children during meals in order to elucidate differences in the calorie and macronutrient content of meals and the circulating hormones related to hunger and satiety in those who either endorse binge-eating behaviors or report no such behaviors. We hypothesize that differences in these factors will predict the development of obesity in the populations studied and may be important for developing rational approaches for the prevention and treatment of obesity in the diverse U.S. population. A new clinical protocol based on a successful pilot study is examining the effects of a targeted interpersonal therapy intervention on body weight change in adolescents who endorse binge-eating behaviors.
Treatment of obesity and the co-morbid conditions associated with obesity
Given the rapid increase in the prevalence of obesity, the development of treatments for obesity in children and adults is urgently needed. In four clinical protocols, we have examined approaches for the prevention and treatment of excessive body weight. We completed a pilot study demonstrating that severely obese adolescents can lose weight when enrolled in a comprehensive weight management program that includes the gastrointestinal lipase inhibitor orlistat as an adjunct to a behavior-modification program. We recently completed a placebo-controlled randomized trial, studying whether the weight loss of African American and Caucasian children and adolescents with obesity-related co-morbidities was improved by the use of orlistat 120 mg TID. Subjects participated in a 12-week weight reduction program. We compared body weight and body composition (by DXA and air displacement plethysmography), glucose homeostasis by frequently sampled intravenous glucose tolerance test (FSIGT), fasting lipids, pulse, and blood pressure before and after treatment. We studied 200 adolescents, 65% female, 61% African American, mean age14.6± SEM 0.10y, BMI 41.7±0.6 kg/m2 (range 27–87 kg/m2), with 85.5% of subjects completing the trial. Adolescents treated with orlistat lost significantly more weight, BMI units, and fat mass. Although pulse and blood pressure decreased significantly during the trial, orlistat treatment did not significantly alter these variables. Similarly, HOMA-IR, SI by FSIGT, Apo B, total and LDL-cholesterol, and triglycerides decreased in proportion to weight loss, but orlistat use was not associated with significant reductions in any of these obesity-related laboratory co-morbidities. Both aspartate aminotransferase (AST) (+1.8±0.9 vs. −1.08±0.9) and alanine aminotransferase (ALT) (+1.3±1.1 versus −2.4±1.2) unexpectedly increased significantly with orlistat treatment. We concluded that orlistat added to a behavioral program significantly improved weight loss over a six-month interval but had little impact on obesity-related co-morbid conditions in obese adolescents.
A second now-completed study (4) examined the mechanism by which metformin may affect the body weight of younger children with hyperinsulinemia and therefore at risk for later development of Type 2 diabetes. We conducted a single-center, six-month, randomized, double-blind, placebo-controlled trial of the effects of metformin, 1,000 mg BID administered with meals, in severely obese children (6–12 years of age) who manifested hyperinsulinemia and insulin resistance. Subjects participated in a monthly dietitian-administered weight reduction program. We measured body mass index and body composition (by air displacement plethysmography), glucose homeostasis (by HOMA-IR), and lipids before and after 6-months' treatment. Between October 2000 and April 2007, we enrolled 100 obese children (60% female, 11% Hispanic, 3% Asian, 40% African American), mean age 10.2±1.5y, with mean BMI 34.6±6.6 kg/m2 (range 23-58 kg/m2), with 85% of subjects (84% given metformin and 86% given placebo) completing the trial. Compared with placebo-treated children, the BMI, BMI-Z score, and body fat mass of those randomized to metformin declined to a significantly greater extent. Serum glucose (P=0.018) and HOMA-IR also decreased more in metformin-treated than in placebo-treated children. We concluded that metformin, added to a monthly behavioral program, significantly improved weight loss and lowered insulin resistance over a six-month interval in severely obese, insulin-resistant children.
A third clinical trial in adults examined the role played by dietary calcium in body weight (5). We randomized 340 overweight (BMI 25 to <30 kg/m2) and obese (BMI ≥30 kg/m2) adults to take calcium carbonate (elemental calcium, 1,500 mg/d, n = 170, or placebo, n = 170) with meals for two years. Seventy-five percent of participants completed the trial (78% received calcium; 73% received placebo). There were no statistically or clinically significant differences between the calcium and placebo groups in change in body weight (difference 0.02 kg), BMI (difference 0.32 kg/m2), or body fat mass (difference 0.39 kg). Parathyroid hormone concentrations decreased in the calcium group compared with the placebo group (difference −0.71 pmol/L). We concluded that dietary supplementation with 1,500 mg/d elemental calcium for two years had no statistically or clinically significant effects on weight in overweight and obese adults. Calcium supplementation is thus unlikely to have clinically significant efficacy as a weight-gain preventive measure in such patients.
A fourth study examined the role played by central nervous system histamine in controlling food intake at meals. Additional studies slated to begin next year will test (i) therapies directed at ameliorating inadequate leptin signaling in conditions marked by leptin receptor hypofunction and (ii) methods to reduce insulin resistance in adolescents at risk for Type 2 diabetes.
Additional Funding
- NIH Clinical Center "Bench to Bedside" Award: FTO and Eating in Absence of Hunger 2009–2011
- Prader-Willi Syndrome Association (USA) Best Idea Grant for Hyperphagia Research
- NIH Clinical Center "Bench to Bedside" Award: Fat Metabolism and Function-Altering Polymorphisms in MC3R 2011–2013
- NIH Clinical Center "Bench to Bedside" Award: Depression and Insulin Resistance in Adolescent Girls 2011–2013
Publications
- Savastano DM, Tanofsky-Kraff M, Han JC, Ning C, Sorg RA, Roza CA, Wolkoff LE, Jefferson-George KS, Figueroa RE, Sanford EL, Brady S, Kozlosky MS, Schoeller DA, Yanovski JA. Energy intake and energy expenditure among children with polymorphisms of the melanocortin-3 receptor. Am J Clin Nutr 2009;90:912-920.
- Han JC, Liu QR, Jones M, Levinn RL, Menzie CM, Jefferson-George KS, Adler-Wailes DC, Sanford EL, Lacbawan FL, Uhl GR, Rennert OM, Yanovski JA. Brain derived neurotrophic factor and obesity in WAGR syndrome. New Engl J Med 2008;359:918-927.
- Tanofsky-Kraff M, Yanovski SZ, Schvey NA, Olsen CH, Gustafson J, Yanovski JA. A prospective study of loss of control eating for body weight gain in children at high risk for adult obesity. Int J Eat Disord 2009;42:26-30.
- Yanovski JA, Krakoff J, Salaita CG, McDuffie JR, Kozlosky M, Sebring NG, Reynolds JC, Brady SM, Calis KA. Effects of metformin on body weight and body composition in obese insulin-resistant children: a randomized clinical trial. Diabetes 2011;60:477-85.
- Yanovski JA, Parikh SJ, Yanoff LB, Denkinger BI, Calis KA, Reynolds JC, Sebring NG, McHugh T. Effects of calcium supplementation on body weight and adiposity in overweight and obese adults: a randomized trial. Ann Intern Med 2009;150:821-829.
Collaborators
- Greti Aguilera, MD, Program on Developmental Endocrinology and Genetics, NICHD, Bethesda, MD
- Leslie Biesecker, MD, GDRB/HDS, NHGRI, Bethesda, MD
- Kong Chen, PhD, Clinical Endocrinology Branch, NIDDK, Bethesda, MD
- Myles Faith, PhD, University of Pennsylvania School of Medicine, Philadelphia, PA
- I. Sadaf Farooqi, MD, Cambridge Institute for Medical Research, Cambridge, UK
- Oksana Gavrilova, PhD, Mouse Metabolism Core Laboratory, NIDDK, Bethesda, MD
- Van S. Hubbard, MD, PhD, Division of Nutritional Research Coordination, NIDDK, Bethesda, MD
- Joel E. Kleinman, MD, PhD, Clinical Brain Disorders Branch, NIMH, Bethesda, MD
- Rudolph L. Leibel, MD, Columbia University College of Physicians and Surgeons, New York, NY
- Stephen O'Rahilly, MD, Cambridge Institute for Medical Research, Cambridge, UK
- Dale A. Schoeller, PhD, University of Wisconsin, Madison, WI
- Marian Tanofsky-Kraff, PhD, USUHS Department of Psychology, Bethesda, MD
- George R. Uhl, MD, PhD, Molecular Neurobiology Branch, NIDA, Baltimore, MD
- B. Timothy Walsh, PhD, Columbia University College of Physicians and Surgeons, New York, NY
- Heiner Westphal, MD, Program on Genomics of Differentiation, NICHD, Bethesda, MD
- Denise E. Wilfley, PhD, Washington University School of Medicine, St. Louis, MO
- Susan Z. Yanovski, MD, Obesity and Eating Disorders Program, NIDDK, Bethesda, MD
Contact
For further information, contact yanovskj@mail.nih.gov or visit ugo.nichd.nih.gov.

