You are here: Home > Section on Growth and Obesity
Physiology, Psychology, and Genetics of Obesity

- Jack A. Yanovski, MD, PhD, Head, Section on Growth and Obesity
- Joan C. Han, MD, Assistant Clinical Investigator
- Bonggi Lee, PhD, Visiting Fellow
- Joo Yun Jun, PhD, Postdoctoral Fellow
- Tania Condarco, MD, Fellow, Adult Endocrine Training Program
- Andrew Demidowich, MD, Fellow, Adult Endocrine Training Program
- Sheila M. Brady, RN, FNP, Nurse Practitioner
- Sarah Berger, PhD, Special Volunteer
- Nichole Kelly, PhD, Research Collaborator
- Rachel Miller-Radin, MA, Research Collaborator
- Anna Vannucci, MA, Research Collaborator
- Robin Roberson, MS, Technician
- Amanda Krause, BS, Postbaccalaureate Fellow
- Dezmond Taylor-Douglas, BS, Graduate Student
The prevalence of overweight and obesity in children and adults has tripled during the past 40 years. The alarming rise in body weight has likely occurred because the current environment affords easy access to calorie-dense foods and requires less voluntary energy expenditure. However, such an environment leads to obesity only in those individuals whose body weight–regulatory systems are not able to control body adiposity with sufficient precision in our high calorie/low activity environment, which suggests there are subgroups in the U.S. with a uniquely high susceptibility to weight gain under the prevailing environmental conditions. Our primary goal is to elucidate the genetic underpinnings of the metabolic and behavioral endo-phenotypes that contribute to the development of obesity in children. Using our unique longitudinal cohort of children at risk for adult obesity, who have undergone intensive metabolic and behavioral phenotyping, we examine genetic and phenotypic factors predictive of progression to adult obesity in children who are in the “pre-obese” state, allowing characterization of phenotypes unconfounded by the impact of obesity itself. Once they are identified as linked to obesity, we study intensively genetic variants that impair gene function. We expect that these approaches will improve our ability to predict which children are at greatest risk for obesity and its comorbid conditions and will lead to more targeted, etiology-based prevention and treatment strategies for pediatric obesity.
Genetic factors important for childhood body weight regulation
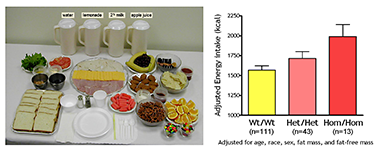
Click image to enlarge.
Figure 1. Energy intake studied by using free-access buffet meals of palatable foods
Children homozygous for two polymorphisms in the MC3R gene (Hom/Hom) consumed more at the buffet than heterozygotes (Het/Het) or those with wild-type MC3R (Wt/Wt).
To identify gene variants affecting body composition, we have been examining polymorphisms in genes involved in the leptin signaling pathway. Genes include FTO (fat mass and obesity-associated gene) and those encoding the leptin receptor, proopiomelanocortin (POMC), the melanocortin 3 receptor (MC3R), brain-derived neurotrophic factor (BDNF), and histaminergic receptors 1 and 3. We are currently studying a variant MC3R that is associated with adiposity in children and appears to have functional significance for MC3R signal transduction. Children who are homozygous variant for both C17A and G241A polymorphisms have significantly greater fat mass and higher plasma levels of insulin and leptin than unaffected or heterozygous children and appear to eat more at laboratory test meals (Figure 1). In vitro studies subsequently found that signal transduction and protein expression were significantly lower for the double mutant MC3R. Our ongoing studies attempt to understand the mechanisms by which these sequence alterations may affect body weight. Transgenic “knock-in” mice expressing the human wild-type and human double-mutant MC3R were therefore developed. Preliminary analyses suggest alterations in body composition consistent with our observations in humans, but provide evidence of surprising metabolic health despite their obesity (Figure 2). We will continue to phenotype the mice intensively over the next two years. We also initiated a clinical investigation of lipolysis and lipogenesis in humans with these polymorphisms.
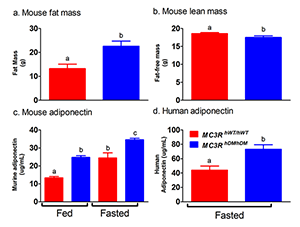
Figure 2. Studies of a human MC3R variant containing two naturally occurring polymorphisms
The variant is associated with pediatric-onset obesity. We found that mice whose MC3R was replaced by human versions of the gene were obese when they expressed the double-mutant gene (MC3RhDM/hDM)—with greater fat mass (panel a) and lower fat-free mass (panel b), but surprisingly greater adiponectin concentrations (panel c) than mice with the normal human MC3R (MC3RhWT/hWT). Humans with the double-mutant receptor also showed greater adiponectin (panel d).
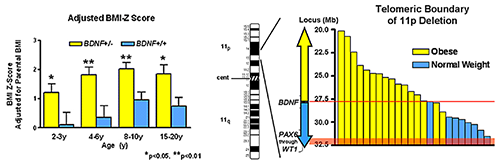
We also investigated the BDNF-TrkB pathway in relation to body mass in children. In a cohort of 328 children, ages 3–19 years, we found obese children had significantly lower BDNF; BMI, BMI-Z, and body fat were all negatively associated with BDNF. The data suggest that some obese individuals with low serum BDNF for age may have mutations that alter BDNF function. We therefore assessed the role of BDNF haploinsufficiency as a cause of obesity in patients with syndromes attributable to deletions in the vicinity of 11p14.1, where the human BDNF gene is located. In 33 subjects with the WAGR (Wilms' tumor, aniridia, genitourinary, and renal abnormalities) syndrome who had heterozygous 11p deletions, ranging in size from 1.0–26.5 Mb, 19 had regions of deletion that involved the BDNF gene (BDNF+/−). Compared with those with intact BDNF (BDNF+/+), BDNF+/− individuals had significantly greater body mass during childhood, starting at age 2 (Figure 3). A recent investigation examined common polymorphisms in the BDNF gene region. We investigated human hypothalamic BDNF expression in association with 44 BDNF single nucleotide polymorphisms (SNPs). We found that the minor C allele of rs12291063 is associated with lower human ventromedial hypothalamic BDNF expression and greater adiposity in both adult and pediatric cohorts. We also demonstrated that the major T allele for rs12291063 binds to the transcriptional regulator heterogeneous nuclear ribonucleoprotein D0B, knockdown of which disrupts transactivation by the T allele. Binding and transactivation functions are both disrupted by substitution of T with the minor C allele. The findings provide a rationale to pursue augmentation of BDNF expression and BDNF receptor signaling as targeted obesity treatments for individuals with the rs12291063 CC genotype.
Figure 3.
Patients with WAGR syndrome who have haploinsufficiency for the brain-derived neurotrophic factor gene (BDNF) had a higher BMI standard deviation score (BMI Z score) than children and adults with WAGR syndrome who retained two copies of the BDNF gene (left panel). Deletions that extended into exon 1 of BDNF were associated with 100% risk of childhood-onset obesity (right panel).
Physiology, metabolism, and psychology of childhood body weight regulation
Our studies are directed at understanding the physiological, psychological, and metabolic factors that place children at risk for undue weight gain. As part of these studies, we examined how best to measure eating-related psychopathology, insulin sensitivity, changes in body composition, energy intake, and energy expenditure in children and we studied the short- and long-term stability of the components of metabolic syndrome. We found that leptin is an important predictor of weight gain in children: those with high leptin levels gain even more weight when followed longitudinally than do children with normal leptin levels. We documented that hyperinsulinemia is positively related to energy intake in non-diabetic, obese children. We also examined the relationship between depressive symptomatology and insulin resistance in children and adolescents, finding strong associations both cross-sectionally and prospectively between depressive symptoms and insulin resistance independent of body weight. The associations suggest mechanisms whereby insulin resistance contributes to excessive weight gain in children and have informed some of our treatment approaches to pediatric obesity (described below).
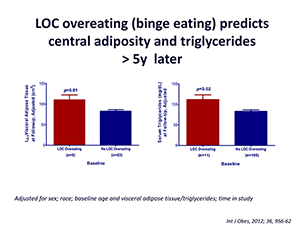
Click image to enlarge.
Figure 4.
On average (±SE), children who engage in binge eating at baseline have more visceral adipose tissue at L2-3 at follow-up than children who do not engage in binge eating at baseline, adjusting for sex, race, baseline age, baseline visceral adipose tissue at L2-3, and time in study (P = 0.01).
On average (±SE), children who engage in binge eating at baseline have higher follow-up triglycerides than children who do not engage in binge eating at baseline, adjusting for sex, race, baseline age, body mass index (kg/m2), baseline triglycerides, and time in study (P = 0.02).
Our evaluations concentrating on binge-eating behaviors in children suggest that such behaviors are also associated with adiposity in children. We also found that binge-eating and dieting behaviors may predict future weight gain in children at risk for obesity: Children reporting binge-eating behaviors such as loss of control (LOC) over eating gained, on average, 2.4 kg of weight more per year than did non-binge-eating children. Our data also suggest that children endorsing binge eating consume more energy during meals. Intake during buffet meals averaged 400 kcal more in children who engage in binge eating than children who do not, but despite their greater intake, such children reported shorter-lived satiety than children without binge-eating episodes. The ability to consume large quantities of palatable foods, especially when coupled with decreased subsequent satiety, may play a role in the greater weight gain found in binge-eating children. Interestingly, we recently demonstrated that, among a cohort of 506 lean and obese youth, youth with LOC over eating had significantly higher serum leptin levels than those without LOC episodes, even after adjusting for adiposity and other relevant covariates (1). The data also suggest that interventions targeting disordered eating behaviors may be useful in preventing excessive fat gain in children prone to obesity and have led to ongoing trials to study preventive strategies related to binge eating.
Because binge eating appears to be a heritable trait, we initiated studies to investigate potential genetic factors linked to LOC over eating. For example, we previously reported that, among 229 youth aged 6–19 y who were genotyped for the FTO single nucleotide polymorphism (SNP) rs9939609, subjects with at least one A (at-risk) allele (67.7%) had significantly elevated BMI, BMI z scores, and fat mass. Of the AA/AT subjects, 34.7% reported LOC over eating compared with 18.2% of the TT (normal alleles) subjects. AA/AT subjects consumed a significantly greater percentage of energy from fat than did the TT subjects. We also found preliminary evidence of a link between FTO SNP rs9939609 and eating in the absence of hunger.
We study normal-weight children and adolescents, children who are already obese, and the non-obese children of obese parents, in order to determine the factors that are most important for development of the complications of obesity in youth. Body composition, leptin concentration, metabolic rate, insulin sensitivity, glucose disposal, energy intake at buffet meals, and genetic factors believed to regulate metabolic rate and body composition are examined. Psychological and behavioral factors, such as propensity to engage in binge-eating behavior (Figure 4), are also studied. Children are being followed longitudinally into adulthood. In two protocols, we study actual food consumption of children during meals, to elucidate differences in the calorie and macronutrient content of meals and the circulating hormones related to hunger and satiety in those who either endorse binge-eating behaviors or report no such behaviors. We found that eating in the absence of physiological hunger is a replicable trait that appears linked to obesity. We hypothesize that differences in these factors will predict the development of obesity in the populations studied and may be of great importance in developing rational approaches for the prevention and treatment of obesity in the diverse U.S. population.
Treatment of obesity and the co-morbid conditions associated with obesity
Given the rapid increase in the prevalence of obesity, the development of treatments for obesity in children and adults is urgently needed, yet current pharmacologic approaches are extremely limited for both children and adults (2). In several clinical protocols, we examined approaches for the prevention and treatment of excessive body weight. We completed a pilot study demonstrating that severely obese adolescents can lose weight when enrolled in a comprehensive weight management program that includes the gastrointestinal lipase inhibitor orlistat as an adjunct to a behavioral modification program. We also completed a placebo-controlled randomized trial, studying whether the weight loss of African American and Caucasian children and adolescents who have obesity-related comorbidities was improved by the use of orlistat 120 mg TID. Subjects participated in a 12-week weight reduction program. Body weight and body composition (by DXA and air displacement plethysmography), glucose homeostasis by frequently sampled intravenous glucose tolerance test (FSIGT), fasting lipids, pulse, and blood pressure were compared before and after treatment. We studied 200 adolescents, 65% female, 61% African American, mean age±SEM 14.6±0.10y, BMI 41.7±0.6 kg/m2 (range 27–87 kg/m2). Of the subjects 85.5% completed the trial. Adolescents treated with orlistat lost significantly more weight, BMI units, and fat mass than those who did not receive the drug. Although pulse and blood pressure declined significantly during the trial, orlistat treatment did not significantly alter these variables. Similarly, HOMA-IR (homeostasis model assessment-insulin resistance), SI (insulin resistance) by FSIGT, apolipoprotein B, total and LDL-cholesterol, and triglycerides declined significantly in proportion to weight loss, but orlistat use was not associated with significant reductions in any of these obesity-related laboratory comorbidities. Unexpectedly, both aspartate-aminotransferase (AST) and alanine-aminotransferase (ALT) increased significantly with orlistat treatment. We concluded that orlistat added to a behavioral program significantly improved weight loss over a 6-month interval, but had little impact on obesity-related co-morbid conditions in obese adolescents. We also took advantage of this trial to study free fatty acid (FFA) flux in obese boys and girls (3). In adolescents, it was unknown whether sex-associated differences in FFA flux occur. We therefore studied FFA kinetics in 112 non-Hispanic white and black adolescents (31% male; age range, 12–18 years; body mass index SD score range, 1.6–3.1). Glucose, insulin, and FFA were measured during insulin-modified frequently sampled i.v. glucose tolerance tests. Minimal models for both glucose and FFA calculated insulin sensitivity index (SI) and FFA kinetics, including maximum (l0 + l2) and insulin-suppressed (l2) lipolysis rates, clearance rate constant (cf), and insulin concentration for 50% lipolysis suppression (ED50). Relationships of FFA measures to sex, resting energy expenditure (REE), fat mass (FM), lean body mass (LBM) and visceral adipose tissue (VAT) were examined. In models accounting for age, race, pubertal status, height, FM, and LBM, we found sex, pubertal status, age, and REE independently contributed significantly to the prediction of l2 and l0 + l2. Sex and REE independently predicted ED50. Sex, FM/VAT, and LBM were independent predictors of cf. Girls had greater l2, l0 + l2 and ED50 and greater cf than boys. Thus, independent of the effects of REE and FM, FFA kinetics differ significantly in obese adolescent girls and boys, suggesting greater FFA flux among girls.
A second obesity treatment study examined the mechanism by which metformin may affect the body weight of younger children who have hyperinsulinemia, and are therefore at risk for later development of type 2 diabetes. We conducted a single-center, 6-month, randomized, double-blind, placebo-controlled trial of the effects of metformin, 1000 mg BID administered with meals, in severely obese children (6–12y) who manifested hyperinsulinemia and insulin resistance. Subjects participated in a monthly dietitian-administered weight reduction program. Body mass index and body composition (by air displacement plethysmography), glucose homeostasis (by HOMA-IR), and lipids were measured before and after 6-months’ treatment. 100 obese children (60% female, 11% Hispanic, 3% Asian, 40% African American), mean age 10.2±1.5y, with mean BMI 34.6±6.6 kg/m2 (range 23–58 kg/m2) were enrolled between October, 2000, and April, 2007. Of the subjects, 85% (84% given metformin and 86% given placebo) completed the trial. Those randomized to metformin reduced BMI, BMI-Z score, and body fat mass to a significantly greater extent than did placebo-treated children. Serum glucose and HOMA-IR also declined more in metformin-treated than in placebo-treated children. We concluded that metformin, added to a monthly behavioral program, significantly improved weight loss and insulin resistance over a six-month interval in severely obese, insulin-resistant children.
A third clinical trial (4) examined the efficacy of two diets among Hispanic children and adolescents, for whom the prevalence of obesity and insulin resistance is considerably greater than for non-Hispanic white children. A low-glycemic load diet (LGD) has been proposed as an effective dietary intervention for pediatric obesity, but no published study had examined the effects of an LGD in obese Hispanic children. We compared the effects of an LGD and a low-fat diet (LFD) on body composition and components of metabolic syndrome in obese Hispanic youth. Obese Hispanic children (7–15 y of age) were randomly assigned to consume an LGD or an LFD in a two-year intervention program. Body composition and laboratory assessments were obtained at baseline and 3, 12, and 24 mo after intervention. In the 113 children who were randomly assigned, 79% of both groups completed three months of treatment, and 58% of LGD and 55% of LFD subjects attended 24-month follow-up. Compared with the LFD, the LGD significantly reduced the glycemic load per kilocalories of reported food intakes in participants at three months. Both groups had a significantly reduced BMI z score, which was expressed as a standard z score relative to CDC age- and sex-specific norms, and improved waist circumference and systolic blood pressure at 3, 12, and 24 months after intervention. However, there were no significant differences between groups for changes in BMI, insulin resistance, or components of metabolic syndrome. We found no evidence that an LGD and an LFD differ in efficacy for the reduction of BMI or aspects of metabolic syndrome in obese Hispanic youth. Both diets reduced the BMI z score when prescribed in the context of a culturally adapted, comprehensive weight-reduction program.
A fourth, recently completed study examined prevention of weight gain using interpersonal therapy in adolescents reporting LOC eating behaviors (5). Girls were randomly assigned to participate in an adapted interpersonal psychotherapy or a health-education group program for 12 weekly 90-min group sessions. Participation in both conditions was associated with decreases in expected BMI gain, age-adjusted BMI metrics, the percentage of fat by using dual-energy X-ray absorptiometry, symptoms of depression and anxiety, and the frequency of loss-of-control eating over 12 mo of follow-up with no group difference. Follow-up assessments occurred immediately after group programs and at 6 and 12 mo. Interpersonal psychotherapy was found to be more efficacious than health education at reducing objective binge eating at the 12-mo follow-up.
An additional ongoing study tests methods to reduce insulin resistance in adolescents at-risk for Type 2 diabetes. The protocol completed accrual in 2014 and will complete follow-up in 2015. A novel intervention to be studied in the next 3 years with a randomized-controlled trial involves use of colchicine to ameliorate the inflammation of obesity and thus reduce its complications.
Additional Funding
- NIH Clinical Center "Bench to Bedside" Award: Testing Neurobehavioral Endophenotypes of Loss of Control Eating, NICHD 2014–2015
- NICHD Division of Intramural Research Director's Investigator Award: Suppression of NLRP3 inflammasome activation to ameliorate obesity-associated metabolic abnormalities 2014–2015 s
- Office of Disease Prevention, NIH: Grant supplement to support the clinical protocol “Mood and Insulin Resistance in Adolescents at Risk for Diabetes” 2014
Publications
- Miller R, Tanofsky-Kraff M, Shomaker LB, Field SE, Hannallah L, Reina S, Mooreville M, Sedaka N, Brady SM, Condarco T, Reynolds JC, Yanovski SZ, Yanovski JA. Serum leptin and loss of control eating in children and adolescents. Int J Obes 2014;38:397-403.
- Yanovski SZ, Yanovski JA. Long-term drug treatment for obesity: a systematic and clinical review. JAMA 2014;311:74-86.
- Adler-Wailes DC, Periwal V, Ali AH, Brady SM, McDuffie JR, Uwaifo GI, Tanofsky-Kraff M, Salaita CG, Hubbard VS, Reynolds JC, Chow CC, Sumner AE, Yanovski JA. Sex-associated differences in free fatty acid flux of obese adolescents. J Clin Endocrinol Metab 2013;98:1676-1684.
- Mirza NM, Palmer MG, Sinclair KB, McCarter R, He J, Ebbeling CB, Ludwig DS, Yanovski JA. Effects of a low glycemic load or a low-fat dietary intervention on body weight in obese Hispanic American children and adolescents: a randomized controlled trial. Am J Clin Nutr 2013;97:276-285.
- Tanofsky-Kraff M, Shomaker LB, Wilfley DE, Young JF, Sbrocco T, Stephens M, Ranzenhofer LM, Elliott C, Brady S, Radin RM, Vannucci A, Bryant EJ, Osborn R, Berger SS, Olsen C, Kozlosky M, Reynolds JC, and Yanovski JA. Targeted prevention of excess weight gain and eating disorders in high-risk adolescent girls: a randomized controlled trial. Am J Clin Nutr 2014;100:1010-1018.
Collaborators
- Leslie Biesecker, MD, Genetic Disease Research Branch, NHGRI, Bethesda, MD
- Andrew Butler, PhD, The Scripps Research Institute, La Jolla, CA
- Kong Chen, PhD, Clinical Endocrinology Branch, NIDDK, Bethesda, MD
- Samuel Cushman, PhD, Diabetes Branch, NIDDK, Bethesda, MD
- Myles Faith, PhD, University of Pennsylvania School of Medicine, Philadelphia, PA
- I. Sadaf Farooqi, MD, Cambridge Institute for Medical Research, Cambridge, UK
- Katherine Flegal, MPH, PhD, National Center for Health Statistics, Centers for Disease Control and Prevention, Hyattsville, MD
- Oksana Gavrilova, PhD, Mouse Metabolism Core Laboratory, NIDDK, Bethesda, MD
- Van S. Hubbard, MD, PhD, Division of Nutritional Research Coordination, NIDDK, Bethesda, MD
- Joel E. Kleinman, MD, PhD, Clinical Brain Disorders Branch, NIMH, Bethesda, MD
- Rudolph L. Leibel, MD, Columbia University College of Physicians and Surgeons, New York, NY
- Stephen O'Rahilly, MD, Cambridge Institute for Medical Research, Cambridge, UK
- Dale A. Schoeller, PhD, University of Wisconsin, Madison, WI
- Lauren B. Shomaker, PhD, University of Colorado, Boulder, CO
- Eric Stice, PhD, Oregon Research Institute, Eugene, OR
- Marian Tanofsky-Kraff, PhD, Uniformed Services University of the Health Sciences, Bethesda, MD
- George R. Uhl, MD, PhD, Molecular Neurobiology Branch, NIDA, Baltimore, MD
- B. Timothy Walsh, PhD, Columbia University College of Physicians and Surgeons, New York, NY
- Heiner Westphal, MD, Program in Genomics of Differentiation, NICHD, Bethesda, MD
- Denise E. Wilfley, PhD, Washington University School of Medicine, St. Louis, MO
- Susan Z. Yanovski, MD, Obesity and Eating Disorders Program, NIDDK, Bethesda, MD
Contact
For further information, contact yanovskj@mail.nih.gov or visit sgo.nichd.nih.gov.