Activity-Dependent Regulation of Neurons by the Neuregulin-ErbB Signaling Pathway

- Andres Buonanno, PhD, Head, Section on Molecular Neurobiology
- Detlef Vullhorst, PhD, Staff Scientist
- Irina Karavanova, PhD, Research Assistant
- Eastman Lewis, PhD, Intramural Research Training Award Postdoctoral Fellow
- Larissa Erben, PhD, Visiting Fellow
- Neha Akella, BS, Postbaccalaureate Fellow
- Jacqueline Welday, BS, Postbaccalaureate Fellow
- Ricardo Murphy, BS, Contractor
Failure of cortical microcircuits to properly regulate excitatory-inhibitory (E-I) balance is a key feature in the etiology of several developmental psychiatric disorders and neurological diseases, such as schizophrenia, autism, ADHD, and epilepsy. E-I balance is important to synchronize the firing pattern of local neuron ensembles, and its dysregulation can degrade cognitive functions and, in extreme cases, result in epileptiform activity. Network activity, in particular oscillatory activity in the gamma-frequency range (30–80 Hz), is altered in psychiatric disorders and may account for their cognitive and behavioral symptoms. We are interested in how Neuregulin and its receptor ErbB4, which are both genetically linked to psychiatric disorders, function in an activity-dependent fashion (i.e., experience) in the developing brain to regulate synaptic plasticity, neuronal network activity, and behaviors that, in rodents, model features of psychiatric disorders. We identified a functional interaction between Neuregulin/ErbB4, glutamatergic receptors (NMDARs), and dopamine (DA) signaling in GABAergic interneurons that is critical for understanding how Neuregulin can regulate E-I balance and synchronous activity in neuronal networks, processes that are important for cognitive functions altered in psychiatric disorders.
Our earlier studies demonstrated that, in the hippocampus and neocortex, expression of ErbB4, the major neuronal Neuregulin receptor, is restricted to GABAergic interneurons, particularly parvalbumin-positive (Pv+), fast-spiking interneurons, which are critically important for modulating gamma oscillation induction and power. Using genetically engineered mouse models, we discovered that Neuregulin–ErbB4 signaling regulates synaptic plasticity, neuronal network activity, and behaviors associated with psychiatric disorders. More recently, our group investigated other aspects of Neuregulin expression throughout the brain, its processing in response to neuronal activity, and its function in distinct neuronal populations of the developing and maturing nervous system. Distinct Neuregulin isoforms originate from four different genes (Neuregulin 1–4) or, in the case of the Neuregulin-1 gene, the use of alternative promoters. Neuregulin mRNAs encode proteins that have either one or two transmembrane (TM) domains. We recently discovered that, contrary to the widely accepted notion that all Neuregulins are targeted to axons to signal in juxtracrine or paracrine fashion, single-pass TM Neuregulins are targeted to the soma of neurons and dual-pass TM Neuregulins are trafficked into axons (Figure 1). Furthermore, we uncovered a novel bidirectional signaling pathway in which single-pass TM Neuregulins are processed and released from the neuronal soma in response to excitatory transmission selectively through NMDARs, and then signal in autocrine fashion by activating ErbB4 receptors on GABAergic interneurons and promote internalization of NMDARs.
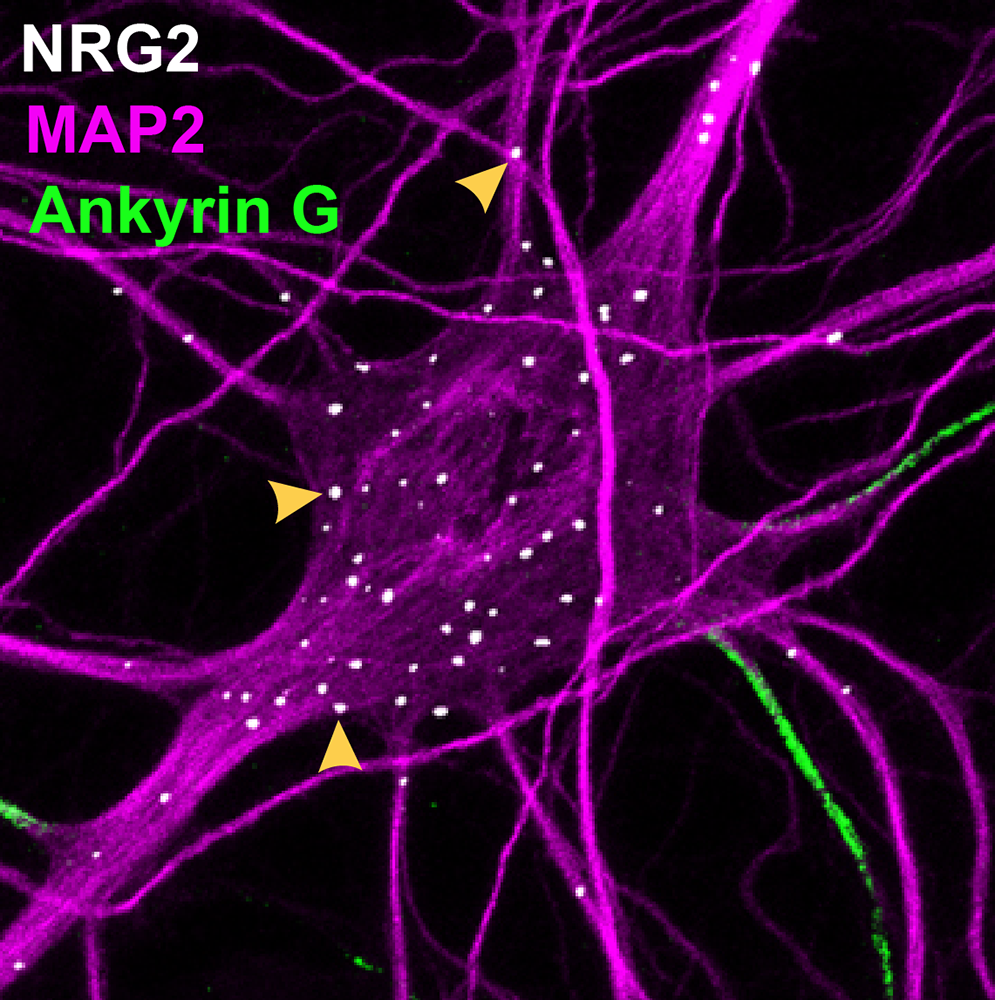
Figure 1. Subcellular NRG2 distribution in a cultured hippocampal neuron
NRG2 (white) accumulates as an unprocessed pro-form on the cell body and proximal dendrites, where it forms puncta at contact sites between the plasma membrane and the endoplasmic reticulum. Axons' initial segments are identified by Ankyrin G (green), dendrites by MAP2 (purple).
Our research uses a combination of in vivo and in vitro techniques that include: optogenetics, fiber photometry combined with genetically encoded calcium (GCaMP) and dopamine (dLight, GRAB) sensors to measure neuronal activity and neurotransmitter release, behavioral analyses that evaluate cognitive performance of wild-type and genetically modified mice using automated touchscreen systems, electrophysiological recordings in acute brain slices, confocal fluorescence microscopy of fixed and live tissues, proteomics analyses, and fluorescence-based multi-channel in situ hybridization (RNAscope, BaseScope). The ultimate goal of such multi-disciplinary approaches is to generate holistic models to investigate the developmental impact of genes that modulate E-I balance and neuronal network activity and that consequently influence behaviors and cognitive functions affected in psychiatric and neurological disorders.
Subcellular distribution and functions of single- vs. dual-transmembrane Neuregulins in central neurons
We recently found that single-pass transmembrane NRGs (sp-NRGs), such as NRG1 type II and NRG2, traffic as unprocessed pro-forms to the neuronal cell surface, where they accumulate at ER–PM junctions on neuronal somata and proximal dendrites. We had found that shedding of the signaling-competent ectodomain of sp-NRGs is triggered by glutamatergic (i.e., excitatory) activity acting on NMDA receptors, and mediated by membrane-bound ADAM–type metalloproteinases. NMDA receptor activity promotes dissociation of unprocessed proNRGs from ER–PM contact sites through dephosphorylation of several conserved Ser/Thr residues located in their intracellular region and by ectodomain shedding by ADAM10 (Figure 2). Together, the two processes promote rapid, regulated release of biologically active sp-NRGs within minutes of NMDA receptor activation to promote ErbB4 signaling [Reference 1].
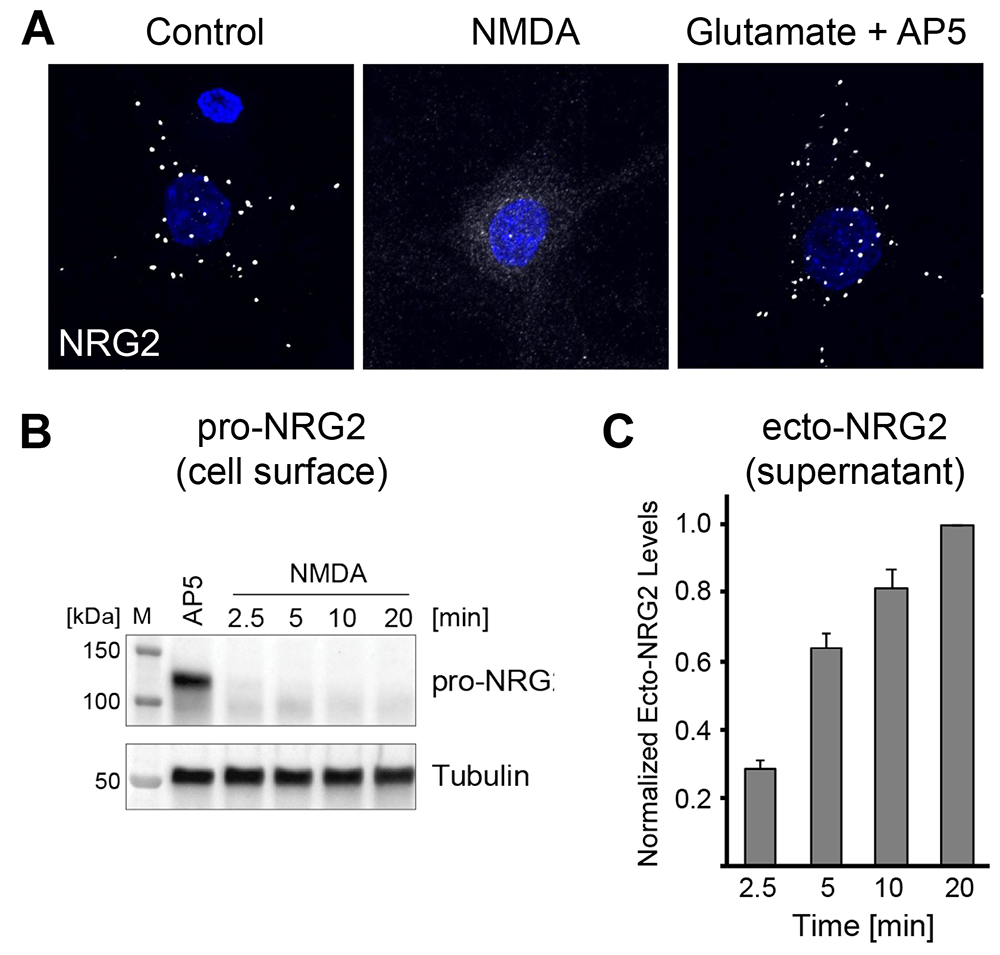
Figure 2. Activity-dependent pro-NRG2 processing
A. Downregulation of NRG2 puncta on cultured hippocampal neurons in response to acute NMDA receptor stimulation.
B. Rapid downregulation of pro-NRG2 protein in whole-cell lysates of cultured neurons treated with 50 µm NMDA for the indicated times.
C. Accumulation of the NRG2 ectodomain in hippocampal culture supernatants following NMDA receptor stimulation, measured by ELISA.
In stark contrast to sp-NRGs, dual-pass transmembrane (dp) NRG1 type III and NRG3 are targeted to axons and accumulate at glutamatergic presynaptic terminals onto GABAergic interneurons, where they signal in juxtacrine mode via postsynaptic ErbB4 receptors. Published and ongoing studies, primarily on NRG3, demonstrate that their unprocessed proforms are cleaved near the second TM domain by BACE-1 (an aspartic acid protease important for the formation of myelin sheaths in peripheral nerve cells), which is required for NRG3 to be released from the Golgi compartment and transported into axons (Figure 3).
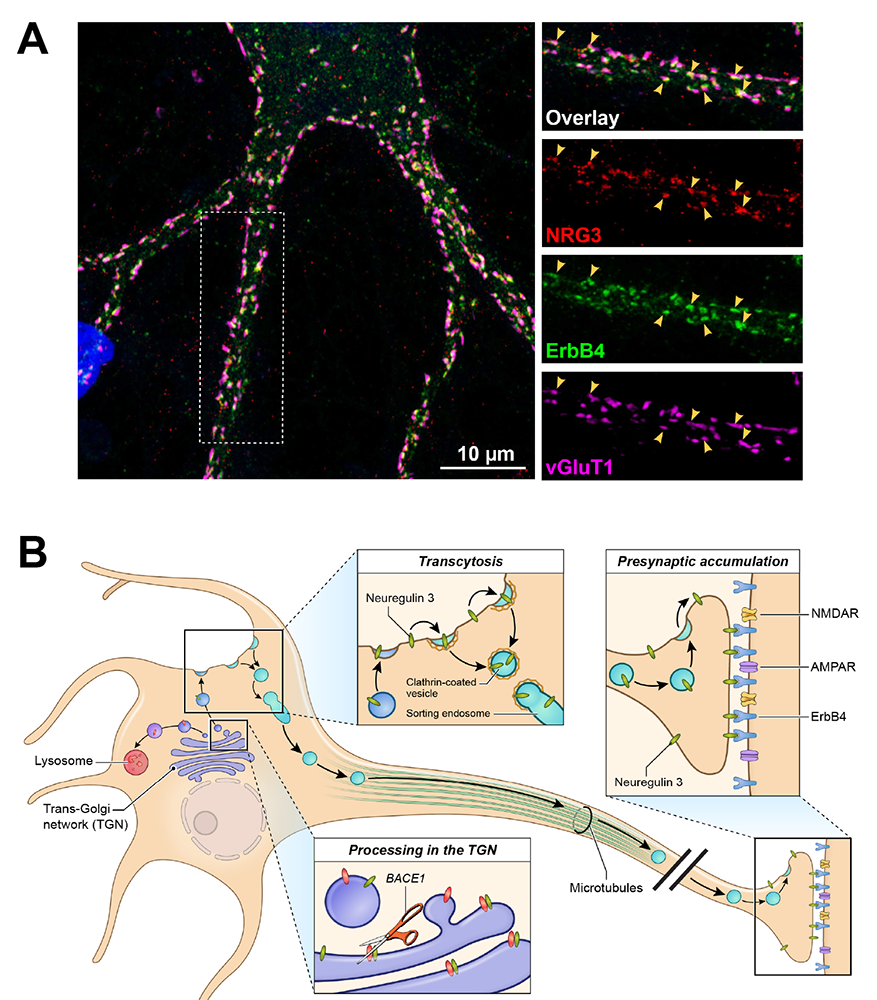
Figure 3. Presynaptic accumulation of NRG3 at glutamatergic presynaptic terminals onto ErbB4+ GABAergic interneurons
A. Immunofluorescence cytochemistry showing co-localization of NRG3, ErbB4, and the vesicular glutamate transporter 1 (vGluT1) on a cultured GABAergic interneuron.
B. Model summarizing the results of our NRG3 trafficking studies. Unprocessed pro-NRG3 is cleaved by the protease BACE1 in the trans-Golgi network (TGN). Processed NRG3 then traffics to the somatodendritic cell surface, from where is re-endocytosed by a process known as transcytosis, sorted, and packaged in Rab4+ vesicles, which are anterogradely transported into axons. NRG3 accumulation at axon terminals is mediated by binding of postsynaptic ErbB4 receptors.
Neuregulin-2 and ErbB4 knockout mice exhibit dopamine dysregulation and severe behavioral phenotypes with relevance to psychiatric disorders.
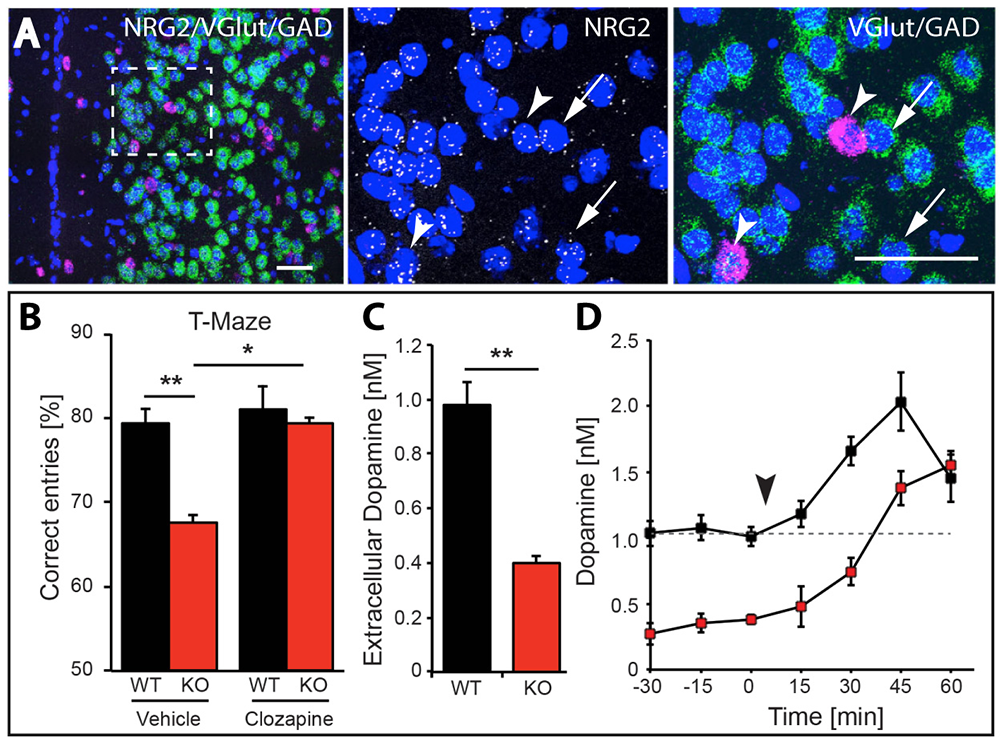
We found that NRG2 expression is more extensive than originally reported and includes striatal and medial prefrontal cortical (mPFC) neurons, while its receptor ErbB4 is expressed on projections from mesencephalic dopamine (DA) neurons that innervate these structures. To investigate the function of NRG2-ErbB4 signaling, we generated knockout (KO) mice for both. We found that NRG2 and ErbB4 KOs have higher extracellular DA levels in the dorsal striatum but lower levels in the mPFC and hippocampus, a pattern similar to DA dysbalance in schizophrenia. NRG2 and ErbB4 KO mice perform abnormally in a battery of behavioral tasks relevant to psychiatric disorders (Figure 4). They exhibit hyperactivity in a novelty-induced open field, deficits in prepulse inhibition, hypersensitivity to amphetamine, antisocial behaviors, reduced anxiety-like behavior in the elevated plus maze, and deficits in the T-maze alternation reward test, a task that is dependent on hippocampal and mPFC function. Acute administration of clozapine, a potent atypical antipsychotic, rapidly increases extracellular DA levels in the mPFC and improves alternation T-maze performance in NRG2 KO mice (Figure 5). The work emphasizes the importance of the NRG2-ErbB4 signaling pathway for proper function of the nigrostriatal, mesocortical, and mesolimbic DA systems [References 2 and 3].
Figure 4. Overlapping behavioral and neurochemical phenotypes in NRG2 and ErbB4 KO mice
Lack of either NRG2 or ErbB4 in genetically engineered mice elicits similar phenotypic alterations with relevance to psychiatric disorders, demonstrating that NRG2 is an important and non-redundant ErbB4 receptor ligand in the postnatal brain.
| NRG2-/- | ErbB4-/- | |
|---|---|---|
| Open Field (Hyperactivity) | increased | increased |
| Elevated Plus Maze (Anxiety) | reduced | reduced |
| Prepulse Inhibition (Sensorimotor Gating) | reduced | reduced |
| T-Maze (Working Memory) | reduced | reduced |
| Amphetamine Sensitivity | increased | increased |
Figure 5. Working memory deficits and reduced dopamine levels in KO mice are temporarily restored by clozapine administration.
A. Expression of NRG2 in the prefontal cortex (PFC) analyzed by triple in situ hybridization. NRG2 transcripts (white) are expressed in both glutamatergic (green) and GABAergic (magenta) neurons.
B. Poor performance by NRG2 KO mice in a T-maze reward alternation task, as compared with WT littermates (left), can be restored by administration of the antipsychotic drug clozapine (right).
C. Reduced extracellular dopamine levels in the mPFC of NRG2 KO mice.
D. Extracellular dopamine levels in the mPFC of NRG2 KO mice rise after clozapine injection (arrowhead) at a time that coincides with improved performance on the T-maze.
Differential analysis of ErbB4 function in mice harboring targeted mutations in either GABAergic or DAergic neurons
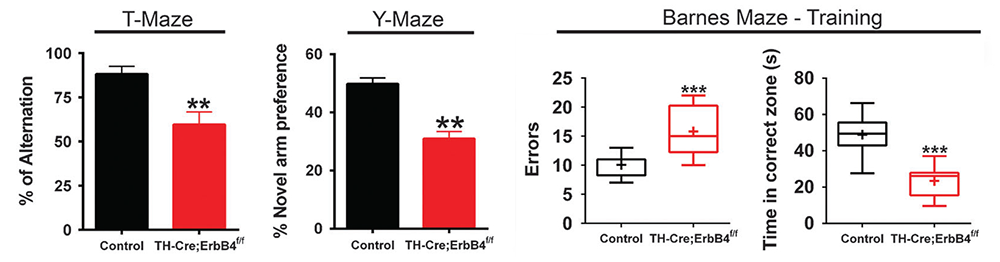
Dysfunctional NRG-ErbB4 signaling in the hippocampus, PFC, and striatum may contribute to alterations in DA function associated with several schizophrenia symptoms. We previously reported that acute injections of NRG1 increase extracellular DA levels and regulate long-term potentiation (LTP) and gamma oscillations, and that ErbB4 is expressed in GABAergic (Pv+) and mesocortical DAergic (TH+) neurons. We therefore used genetic, biochemical, and behavioral approaches to measure DA function in the hippocampus, PFC, and striatum in mice harboring targeted mutations of ErbB4 in either Pv+ or TH+ neurons. Interestingly, we found that, in contrast to GABAergic neurons, ErbB4 is expressed DA neuron axons, and that NRG regulates extracellular DA levels by modulating dopamine transporter (DAT) function. In contrast to mice lacking ErbB4 in Pv+ GABAergic interneurons, which show sensory-motor gating deficits and increases in motor activity, mice lacking ErbB4 in TH+ DAergic neurons exhibit deficits in cognitive-related tasks in the T-, Y- and Barnes- mazes (Figure 6). Therefore, direct effects of NRG/ErbB4 signaling in GABAergic vs. DAergic neurons differentially affect cortical circuits and DA homeostasis and behaviors relevant to schizophrenia [Reference 4].
Figure 6. Working memory deficits in mice lacking ErbB4 in DAergic neurons
Mice were assessed in three different working memory tasks (T-, Y-, and Barnes maze) and were found to perform significantly less well than their control littermates. By contrast, mice lacking ErbB4 in PV+ GABAergic interneurons perform normally in these tasks (not shown).
A novel ultrasensitive in situ hybridization (ISH) approach to detect short sequences and splice variants with cellular resolution
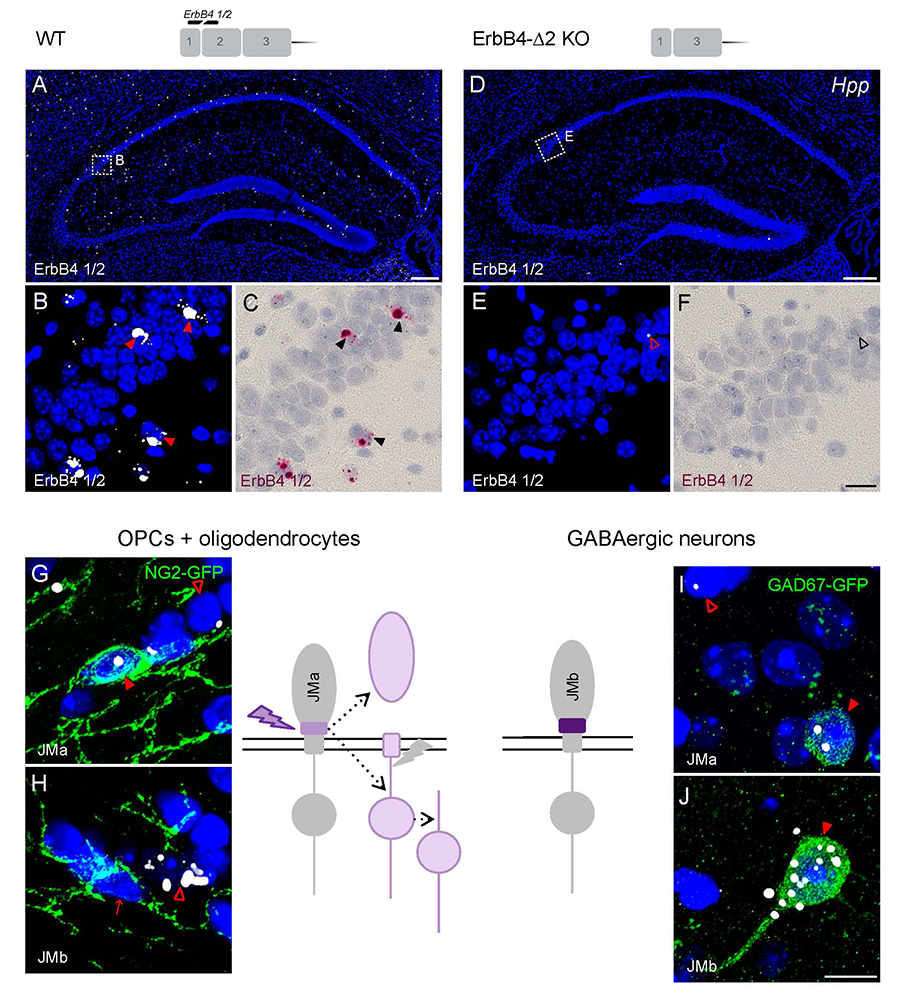
Detection of short isoform-specific sequences typically requires RNA isolation for RT-PCR analysis, an approach that is not suitable to study their regional and cell-type–specific distribution. However, the ability to distinguish differential expression of RNA variants in tissue is highly desirable because alterations in mRNA splicing and editing, as well as coding single-nucleotide polymorphisms, have been associated with numerous cancers as well as with neurological and psychiatric disorders. We reported on a novel highly specific and sensitive single-probe colorimetric/fluorescent ISH approach called BaseScope, which targets short exon/exon RNA splice junctions using single-pair oligonucleotide probes (50 bp), and which differs in several ways from the more traditional approach of RNAscope [Reference 6]. We used BaseScope to investigate, with single-cell resolution, the expression of four ErbB4 transcripts that arise from alternative splicing of exons encoding two juxtamembrane (JMa/JMb) and two cytoplasmic (Cyt-1/Cyt-2) domains (Figure 7). First, we validated the specificity and sensitivity of the approach by comparing ErbB4 hybridization with an exon 2–specific probe on brain sections from wild-type (WT) and ErbB4 KO mice lacking the exon 2. Next, we demonstrated that expression of ErbB4 JM/Cyt isoforms differs between neurons and oligodendrocytes. Based on these results, we propose that BaseScope could serve as an invaluable diagnostic tool to detect alternatively spliced isoforms, and potentially single-base polymorphisms, associated with disease [Reference 5].
Figure 7. BaseScope analysis demonstrates that oligodendrocytes and neurons express distinct ErbB4 juxtamembrane (JM) transcripts.
The sensitivity and specificity of single-pair probe in situ hybridization was demonstrated by the presence of signals in sparse GABAergic hippocampal neurons of WT mice hybridized with a probe corresponding to exon 2 of the ErbB4 gene (A–C), and by the absence of signal in sections prepared from ErbB4 KO mice that lack exon 2 (D–F). Oligodendrocyte precursor cells (OPCs) and mature oligodendrocytes express ErbB4 JMa isoforms, which are susceptible to shedding and back-signaling (G,H), whereas GABAergic neurons express the cleavage-resistant JMb ErbB4 receptor (I,J).
Transduction of mesencephalic DAergic neurons via AAV-Cre microinjection can lead to off-target toxicity.
Stereotaxic microinjection of adeno-associated virus (AAV) expressing Cre recombinase (AAV–Cre) into specific brain regions has become a popular approach to ablate genes by loxP/Cre recombination in a regionally and temporally specific fashion. As part of our investigations into the functional role of the ErbB4 Cyt-1 isoform in DAergic neurons, we microinjected AAV–Cre into the ventral tegmental area (VTA) of mice harboring a conditional (floxed) ErbB4 Cyt-1 allele, using AAV–Cre titers commonly reported in the literature (about 1013 genome copies [GC]/mL). To our surprise, we found that this concentration is toxic to mesencephalic DA neurons. We furthermore found that the toxicity is independent of ErbB4 deletion, as AAV–Cre microinjection in both ErbB4 Cyt-1fl/fl and WT C57Bl/6J mice resulted in similar losses of DA transporter (DAT) and tyrosine hydroxylase (TH) immunoreactivity, proteins critical for DA function, and elicited behavioral changes known to be associated with loss of DA neurotransmission. Furthermore, such phenotypes were observed with different AAV serotypes and proteins expressed (Cre/GFP mixture or Cre-GFP fusion protein). Interestingly, we observed that the AAV1 serotype affects DAT and TH expression, whereas AAV9 is toxic to DAergic neurons when used at about 1013 GC/mL. Importantly, we found that diluting the viral titer 1000-fold to 1010 GC/mL effectively prevents the undesired effects while still efficiently recombining floxed targets. The work highlights the critical need for thorough and appropriate controls, such as including WT mice in experimental design, to account for potential off-target effects when working with AAV–Cre recombinase.
Additional Funding
- Bench-to-Bedside Award (NHD15003-001-00001) “Selective dopamine D4 receptor-targeting compounds as pro-cognitive drugs”
- NICHD DIR Director's Investigator Award (RRC# D-14-07). PI: Andres Buonanno; Co-PI: Juan Bonifacino. “Exploring the functional role of Neuregulin isoform diversity in CNS”
- Center for Compulsive Behaviors Fellowship Award
Publications
- Vullhorst D, Buonanno A. NMDA receptors regulate Neuregulin 2 binding to ER-PM junctions and ectodomain release by ADAM10. Mol Neurobiol 2019;56:8345-8363.
- Yan L, Shamir A, Skirzewski M, Leiva-Salcedo E, Kwon OB, Karavanova I, Paredes D, Malkesman O, Bailey KR, Vullhorst D, Crawley JN, Buonanno A. Neuregulin-2 ablation results in dopamine dysregulation and severe behavioral phenotypes relevant to psychiatric disorders. Mol Psychiatry 2018;23:1233-1243.
- Skirzewski M, Cronin ME, Murphy R, Fobbs W, Kravitz AV, Buonanno A. ErbB4 null mice display altered mesocorticolimbic and nigrostriatal dopamine levels as well as deficits in cognitive and motivational behaviors. eNeuro 2020;7(3):0395-19.2020.
- Skirzewski M, Karavanova I, Shamir A, Erben L, Garcia-Olivares J, Shin JH, Vullhorst D, Alvarez VA, Amara SG, Buonanno A. ErbB4 signaling in dopaminergic axonal projections increases extracellular dopamine levels and regulates spatial/working memory behaviors. Mol Psychiatry 2018;23:2227-2237.
- Erben L, He MX, Laeremans A, Park E, Buonanno A. A novel ultrasensitive in situ hybridization approach to detect short sequences and splice variants with cellular resolution. Mol Neurobiol 2018;55:6169-6181.
- Erben L, Buonanno A. Detection and quantification of multiple RNA sequences using emerging ultrasensitive fluorescent in situ hybridization techniques. Curr Protoc Neurosci 2019;87(1):e63.
Collaborators
- Veronica Alvarez, PhD, Section on Neuronal Structure, NIAAA, Rockville, MD
- Susan G. Amara, PhD, Laboratory of Molecular and Cellular Neurobiology, NIMH, Bethesda, MD
- Juan Bonifacino, PhD, Section on Intracellular Trafficking, NICHD, Bethesda, MD
- Jung Hwa (Susan) Cheng, PhD, EM Facility, NINDS, Bethesda, MD
- Jennie Garcia-Olivares, PhD, Laboratory of Molecular and Cellular Neurobiology, NIMH, Bethesda, MD
- Mario A. Penzo, PhD, Unit on the Neurobiology of Affective Memory, NIMH, Bethesda, MD
- Jung-Hoon Shin, PhD, Section on Neuronal Structure, NIAAA, Rockville, MD
- Hugo Tejeda, PhD, Unit on Neuromodulation and Synaptic Integration, NIMH, Bethesda, MD
- Judith R. Walters, PhD, Experimental Therapeutics Branch, NINDS, Bethesda, MD
Contact
For more information, email buonanno@mail.nih.gov or visit http://smn.nichd.nih.gov.