Activity-Dependent Regulation of Neurons by the Neuregulin-ErbB Signaling Pathway

- Andres Buonanno, PhD, Head, Section on Molecular Neurobiology
- Detlef Vullhorst, PhD, Staff Scientist
- Irina Karavanova, PhD, Research Assistant
- Sonu Singh, PhD, Visiting Fellow
- Miguel Skirzewski, PhD, Visiting Fellow
- Eric Starr, PhD, Postdoctoral Intramural Research Training Award Fellow
- Larissa Erben, MS, Graduate Student
- Marie Cronin, BA, Postbaccalaureate Intramural Research Training Award Fellow
- Ricardo Murphy, BS, Contractor
Failure of cortical microcircuits to properly regulate excitatory-inhibitory (E–I) balance is a key feature in the etiology of several developmental psychiatric disorders and neurological diseases, such as schizophrenia, autism, ADHD, and epilepsy. E–I balance is important to synchronize the firing pattern of local neuron ensembles, and its dysregulation can degrade cognitive functions and, in extreme cases, result in epileptiform activity. Network activity, in particular oscillatory activity in the gamma frequency range (30–80 Hz), is altered in psychiatric disorders and may account for their cognitive and behavioral symptoms. Our lab is interested in how Neuregulin and its receptor ErbB4, which are both genetically linked to psychiatric disorders, function in an activity-dependent fashion (i.e., experience) in the developing brain to regulate synaptic plasticity, neuronal network activity, and behaviors that model features of psychiatric disorders in rodents. We identified a functional interaction between Neuregulin/ErbB4, glutamatergic (NMDA receptors), and dopamine signaling in GABAergic interneurons that is critical for understanding how Neuregulin can regulate E–I balance and synchronous activity in neuronal networks, both processes being important for cognitive functions altered in psychiatric disorders.
Our earlier studies demonstrated that, in the hippocampus and neocortex, expression of ErbB4, the major Neuregulin neuronal receptor, is restricted to GABAergic interneurons, particularly the parvalbumin-positive (Pv+) fast spiking interneurons, which are critically important for modulating gamma oscillation induction and strength (i.e., power). Using genetically targeted mouse models, we discovered that Neuregulin–ErbB4 signaling regulates synaptic plasticity, neuronal network activity, and behaviors associated with psychiatric disorders. More recently our group has been investigating other aspects of Neuregulin expression throughout the brain, its processing in response to neuronal activity, and its function in distinct neuronal populations of the developing and maturing nervous system. Distinct Neuregulin isoforms originate either from four different genes (Neuregulins 1–4) or, in the case of the Neuregulin-1 gene, the use of alternate transcriptional promoters. These mRNAs encode proteins that have either one or two transmembrane (TM) domains. We recently discovered that, contrary to the prior notion that all Neuregulins are targeted to axons to signal in juxtracrine or paracrine fashion, single–TM Neuregulins are targeted to the soma of neurons while dual–TM Neuregulins are trafficked into axons. Interestingly, we uncovered a novel bidirectional signaling pathway in which single–TM Neuregulins are processed and released from neuronal soma in response to excitatory transmission selectively through NMDARs; following release, Neuregulins signal in autocrine fashion by activating ErbB4 receptors on GABAergic interneurons and promote internalization of NMDARs.
Research in our laboratory uses a combination of experimental approaches, including electrophysiological recordings in acute brain slices prepared from normal and genetically altered mice, multi-electrode field recordings from brains of freely moving rats, reverse-microdialysis neurochemistry, confocal fluorescence microscopy in fixed and live tissue, proteomics analyses, and behavioral testing. The ultimate goal of this multi-disciplinary approach is to generate holistic models to investigate the developmental impact of genes that modulate E–I balance and neuronal network activity, and that consequently affect behaviors and cognitive functions altered in psychiatric and neurological disorders.
Click image to view.
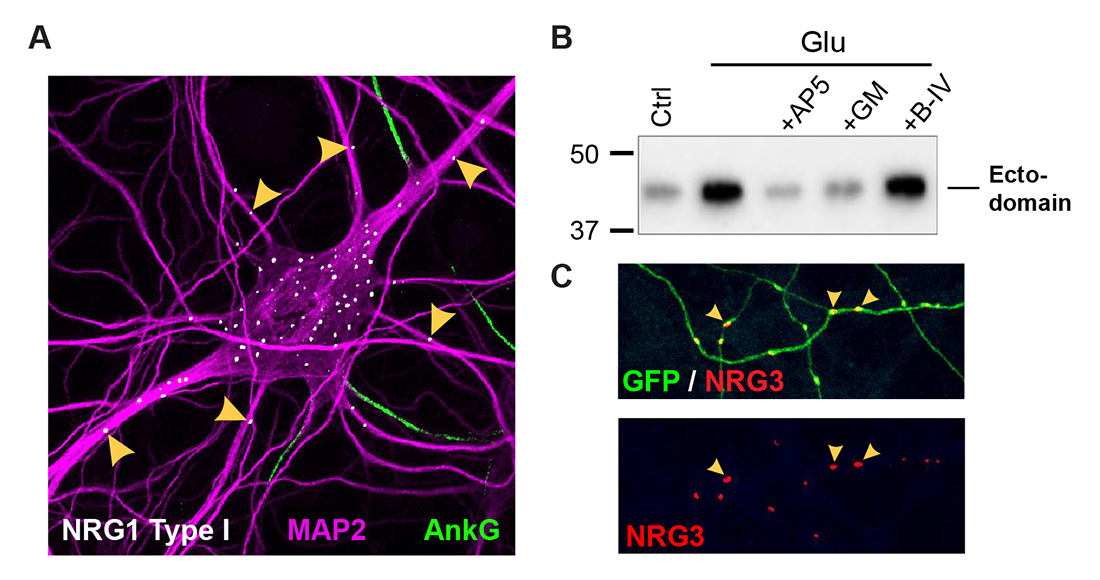
Figure 1. Differential trafficking of NRG isoforms in transfected hippocampal neurons
A. NRG1 type I accumulates as puncta on soma and proximal MAP2–positive dendrites, while axons (proximal segments identified by Ankyrin G) and more distal dendrites are negative.
B. Stimulation of neurons with 20 μM glutamate (Glu) promotes NRG1 ectodomain shedding. The effect is blocked by NMDA receptor (AP5) or metalloprotease (GM6001) inhibition.
C. NRG3 puncta accumulate on GFP–filled axons, away from cell bodies.
Subcellular distribution and functions of single- vs. dual-transmembrane (TM) Neuregulins in central neurons
Numerous Neuregulins (NRGs) are generated through the use of four different genes (NRG1–NRG4) and promoters (NRG1: types -I, -II and -II)), but the functional significance of this evolutionarily conserved diversity remains poorly understood. As summarized below, we recently discovered that NRGs can be categorized into two groups based on their distinct transmembrane (TM) topologies, which impart different subcellular trafficking properties. Using RNAScope and newly generated monoclonal antibodies, we found that NRG2 mRNA and protein are prominently expressed in the developing and adult ErbB4–positive GABAergic interneurons, suggesting that NRG2 can engage in autocrine ErbB4 signaling. Interestingly, we found no evidence of NRG2 protein in axons. Instead, we found that unprocessed pro-NRG2 accumulates at endoplasmic reticulum–plasma membrane (ER–PM) junctions in neuronal soma and proximal dendrites of GABAergic interneurons. Our more recent studies on the other single–TM NRGs (NRG1 types I and II) demonstrate a similar subcellular distribution (Figure 1). Moreover, we found that the ectodomains of all single–TM NRGs are cleaved by alpha-secretases in an activity-dependent manner and shed from the cell surface to signal in paracrine/autocrine fashion. We identified a novel bidirectional signaling pathway in cortical GABAergic interneurons, in which NMDA receptor activation promotes pro-NRG2 shedding and binding to cell-surface ErbB4 receptors, and, in turn, activation of ErbB4 promotes its association with NMDARs and the internalization of these complexes. Consequently, in this fashion, bidirectional signaling between the NRG/ErbB4 and NMDAR pathways function as a homeostatic mechanism to regulate GABAergic interneuron excitatory drive and intrinsic excitatory properties [Vullhorst D, et al., Nat Commun 2015;6:7222]. By contrast to single–TM NRGs, we found that the following dual–TM domain NRGs are targeted to axons, where they signal in juxtacrine mode: CRD (cysteine-rich domain)–NRG1 (type III) and NRG3 (our recent studies uncovered it also is a dual–TM NRG). The findings reveal a previously unknown functional relationship between transmembrane topology, subcellular targeting, and processing (Figure 2), and suggest that single– and dual–TM NRGs regulate neuronal functions in fundamentally different ways [Reference 1]. This work was supported by a Directors Investigator Award.
Click image to view.
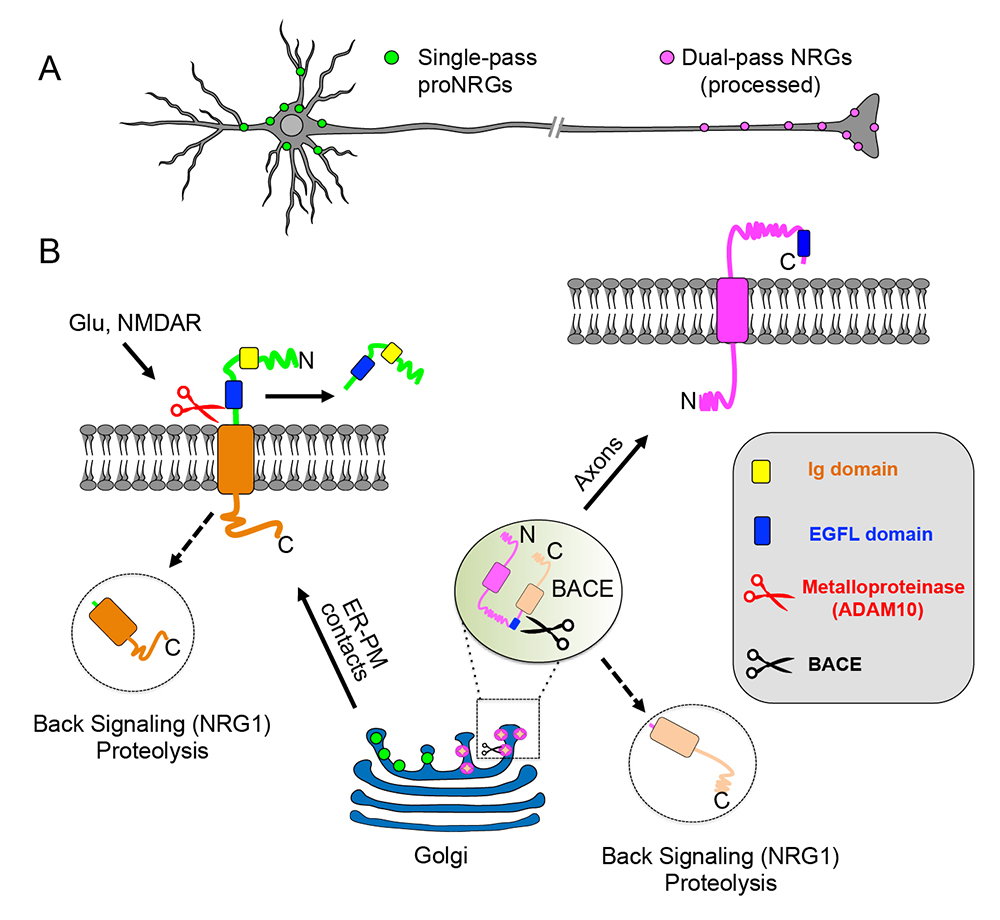
Figure 2. Transmembrane (TM) topology of unprocessed pro–NRGs determines their neuronal subcellular targeting and processing in central neurons.
A. A schematic presentation, based on our findings [References 1 & 2], that depicts single TM NRGs accumulating at somatic subsurface cistern and at proximal dendrites to mediate NRG\ErbB4 signaling, whereas dual TM NRGs accumulate on axons and their terminals to drive long-range signaling.
B. Single TM NRGs are trafficked through the secretory pathway as unprocessed pro-forms and accumulate at subsurface cisternae (SSC) sites, where NMDAR activity promotes ectodomain shedding. By contrast, unprocessed dual TM NRGs traffic via the Golgi and are cleaved by BACE (beta-secretase) to release the amino-terminal region, which is transported into axons in an anterograde manner. Both single– and dual–TM NRGs are processed by gamma-secretase to release carboxyl-terminal proteolytic fragments, which can “back-signal” into the nucleus to regulate transcription.
NMDA receptors regulate NRG2 binding to ER–PM junctions and ectodomain release by ADAM10.
We investigated the temporal and downstream targets of NMDAR activity that mediate pro-NRG2 processing in hippocampal neurons. Using pharmacological and genetic approaches, we found that activation of NMDARs causes the dissociation of single-TM pro-NRGs from SSCs (sub-surface cisternae)/ER–PM junctions and subsequent processing by the sheddase ADAM10 but not by ADAM17, as initially reported in studies using nonneuronal cell lines. Concomitantly, we observed the dephosphorylation in a conserved Ser/Thr–rich region in the intracellular domain of pro-NRG2. Most of the biologically active NRG1 (type-I or -II) or NRG2 ectodomains are shed into the extracellular space within minutes of NMDAR activation, which can then promote signaling via ErbB4 receptors [Reference 2].
A novel ultrasensitive in situ hybridization (ISH) approach to detect short sequences and splice variants with cellular resolution
Detection of short isoform-specific sequences requires RNA isolation for PCR analysis, an approach that loses the regional and cell type–specific distribution of isoforms. The ability to distinguish the differential expression of RNA variants in tissue is critical, because alterations in mRNA splicing and editing, as well as coding single nucleotide polymorphisms, have been associated with numerous cancers and neurological and psychiatric disorders. We reported a novel, highly specific, and sensitive single-probe colorimetric/fluorescent ISH approach, called BaseScope, that targets short exon/exon RNA splice junctions using single-pair oligonucleotide probes (50 bp). We used this approach to investigate, with single-cell resolution, the expression of four ErbB4–encoding transcripts that differ by alternative splicing of exons encoding two juxtamembrane (JMa/JMb) and two cytoplasmic (CYT-1/CYT-2) domains. First, by comparing ErbB4 hybridization on sections from wild-type and ErbB4 knockout mice (missing exon 2), we demonstrated that single-pair probes have the specificity and sensitivity to visualize and quantify the differential expression of ErbB4 isoforms. Next, we demonstrated that expression of ErbB4 isoforms differs between neurons and oligodendrocytes (Figure 3). BaseScope could serve as an invaluable diagnostic tool to detect alternative spliced isoforms, as well as single-base polymorphisms, associated with disease [Reference 3].
Click image to view.
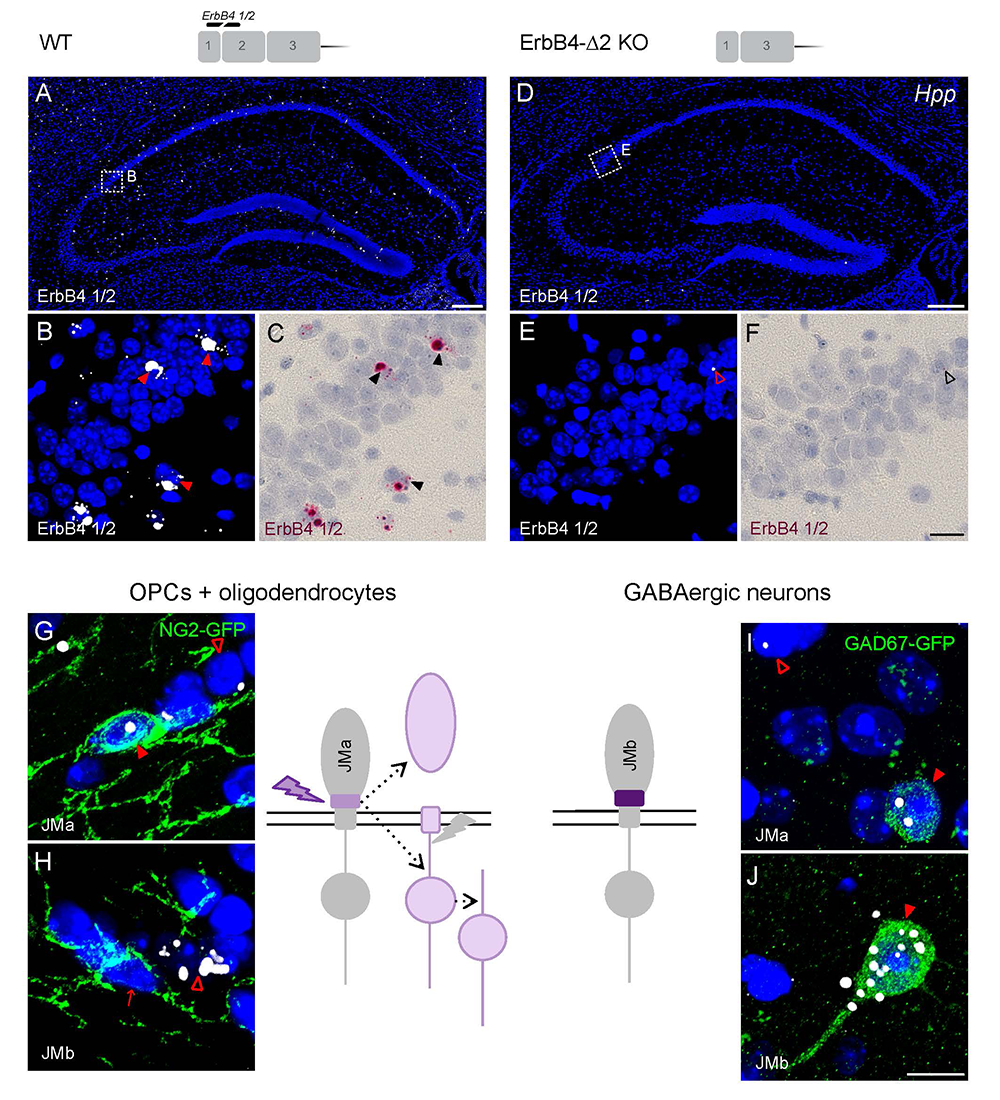
Figure 3. Oligodendrocytes and neurons express distinct ErbB4 JM (juxtamembrane) transcripts.
The sensitivity and specificity of single-pair probe in situ hybridization was demonstrated by the presence of signals in sparse GABAergic hippocampal neurons of wild-type (WT) mice hybridized with a probe corresponding to exon 2 of the ErbB4 gene (A–C), and by the absence of signal in sections prepared from ErbB4 knockout mice that lack exon 2 (D–F). Oligodendrocyte precursor cells (OPCs) and mature oligodendrocytes express ErbB4 JMa isoforms, which are susceptible to shedding and back-signaling (G,H), whereas GABAergic neurons express the cleavage-resistant JMb ErbB4 receptor (I,J).
Neuregulin-2 knockout mice exhibit dopamine dysregulation and severe behavioral phenotypes with relevance to psychiatric disorders.
We found that NRG2 expression in the adult rodent brain does not overlap with NRG1 and is more extensive than originally reported, including expression in the striatum and medial prefrontal cortex (mPFC). We therefore generated NRG2 knockout mice (KO) to study its function. NRG2 KOs have higher extracellular dopamine levels in the dorsal striatum but lower levels in the mPFC, a pattern with similarities to the dopamine dysbalance in schizophrenia. Like ErbB4 KO mice, NRG2 KO mice performed abnormally in a battery of behavioral tasks relevant to psychiatric disorders. They exhibit hyperactivity in a novelty-induced open field, deficits in prepulse inhibition, hypersensitivity to amphetamine, antisocial behaviors, reduced anxiety-like behavior in the elevated plus maze, and deficits in the T-maze alternation reward test, a task that is dependent on hippocampal and mPFC function. Acute administration of clozapine rapidly increased extracellular dopamine levels in the mPFC and improved alternation T-maze performance (Figure 4). Similar to mice treated chronically with N-methyl-D-aspartate receptor (NMDAR) antagonists, we demonstrated that NMDAR synaptic currents in NRG2 KOs are augmented at hippocampal glutamatergic synapses and are more sensitive to ifenprodil, indicating an increased contribution of GluN2B–containing NMDARs. Our findings reveal a novel role for NRG2 in the modulation of behaviors with relevance to psychiatric disorders [Reference 4].
Click image to view.
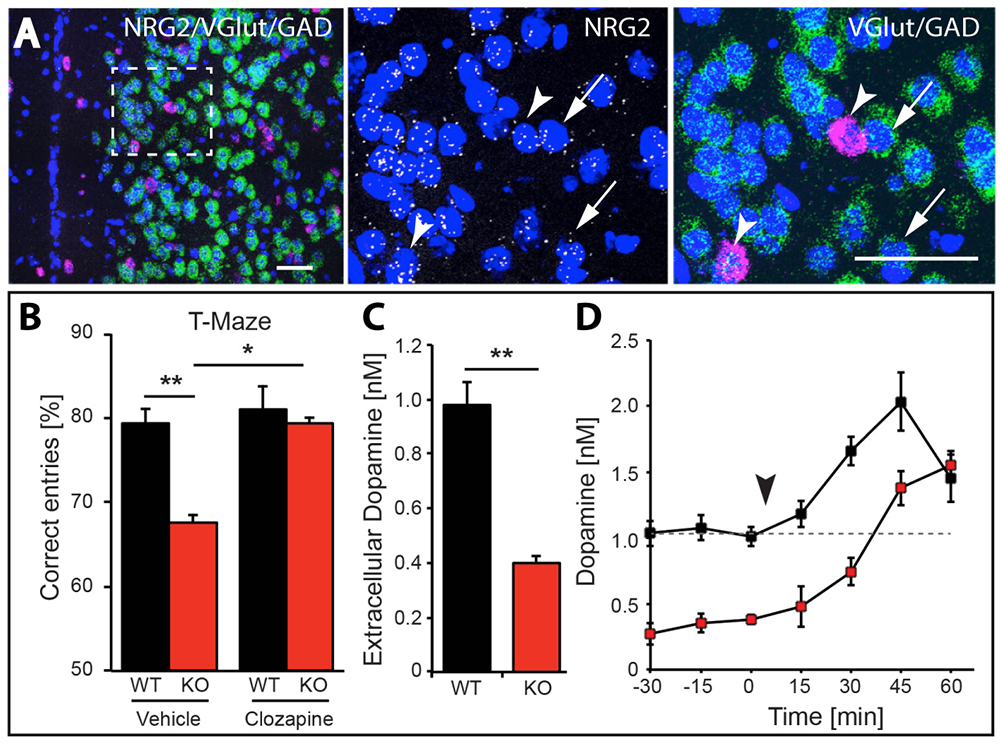
Figure 4. NRG2 null mice exhibit working memory deficits and reduced dopamine levels, which are restored by clozapine.
A. Expression of NRG2 in the prefontal cortex (PFC) analyzed by triple in situ hybridization. NRG2 transcripts (white) are expressed in both glutamatergic (green) and GABAergic (magenta) neurons.
B. Poor performance by NRG2 knockout (KO) mice in a T-maze reward alternation task, as compared with WT littermates (left), can be restored by administration of the antipsychotic drug clozapine (right).
C. Reduced extracellular dopamine levels in the mPFC of NRG2 KO mice.
D. Extracellular dopamine levels in the mPFC of NRG2 KO mice rise after clozapine injection (arrowhead) at a time that coincides with improved performance on the T-maze.
Analysis of ErbB4 function in mice harboring targeted mutations in GABAergic and dopaminergic neurons
Dysfunctional NRG-ErbB4 signaling in the hippocampus, pre-frontal cortex (PFC), and striatum may contribute to alterations in dopamine (DA) function associated with several schizophrenia symptoms. Because NRG1 acutely increases extracellular DA levels and regulates LTP (long-term potentiation) and gamma oscillations, and ErbB4 is expressed in GABAergic (Pv+) and mesocortical DAergic (TH+) neurons, we used genetic, biochemical, and behavioral approaches to measure DA function in the hippocampus, PFC, and striatum of mice harboring targeted mutations of ErbB4 in either PV+ or TH+ neurons. Unexpectedly, we found that, in contrast to GABAergic neurons, ErbB4 is expressed in DA neuron axons, and that NRG regulates extracellular DA levels by modulating DA transporter (DAT) function. In contrast to mice harboring mutations in GABAergic neurons, which show sensorimotor gating deficits and increases in motor activity, ErbB4 TH KO mice exhibit deficits in cognitive-related tasks (in the T-, Y- and Barnes- mazes). Therefore, NRG/ErbB4 signaling in GABAergic vs. DAergic neurons differentially and directly affects cortical circuits and DA homeostasis, as well as behaviors relevant to schizophrenia [Reference 5].
Effects of ketamine on cortical gamma oscillations and the role of dopamine receptors in behaviors
Mounting evidence suggests that gamma oscillations are atypically high at baseline in disorders that affect attention, such as schizophrenia and ADHD (attention deficit/hyperactivity disorder). Ketamine, an antagonist of the NMDAR, has profound effects on gamma oscillation power and phenocopies schizophrenia by eliciting psychoto-mimetic symptoms and affecting cognitive functions in healthy individuals. In collaboration with Judith Walters’s lab, we used multi-electrode recordings from the medial prefrontal cortex (mPFC) and mediodorsal thalamus (MD) of rats acutely treated with ketamine, which serves as a rodent model with “face validity” for schizophrenia, to study the drug’s effects on spiking and gamma local field potentials in the mPFC and MD of freely moving rats. We found that ketamine raises gamma local field potentials and frequencies in both brain areas, but does not increase thalamocortical synchronization. Based on our prior in vitro studies, showing that a “cross-talk” between the dopamine D4 receptor (D4R) and ErbB4 regulates gamma oscillation power in acute hippocampal slices, we investigated whether and how D4R–targeting drugs regulate gamma oscillations. We found that a D4R agonist (A-412997) increased ketamine-induced gamma power, which was blocked by a D4R–selective antagonist (L-745870) in both mPFC and MD, but that neither drug altered ketamine-induced gamma power or frequency in the mPFC (Figure 5). Interestingly, in the MD, the D4R agonist increased the power of ketamine-induced gamma oscillations. Experiments are in progress to evaluate the effect of mutating D4R, in conjunction with pharmacological targeting of D4R receptors, on behaviors that pertain to deficits reported in patients with schizophrenia. We are using the 5-Choice Serial Reaction Time Task (5CSRTT) to quantify the effect of the dopamine receptors on measures of attention and impulsivity.
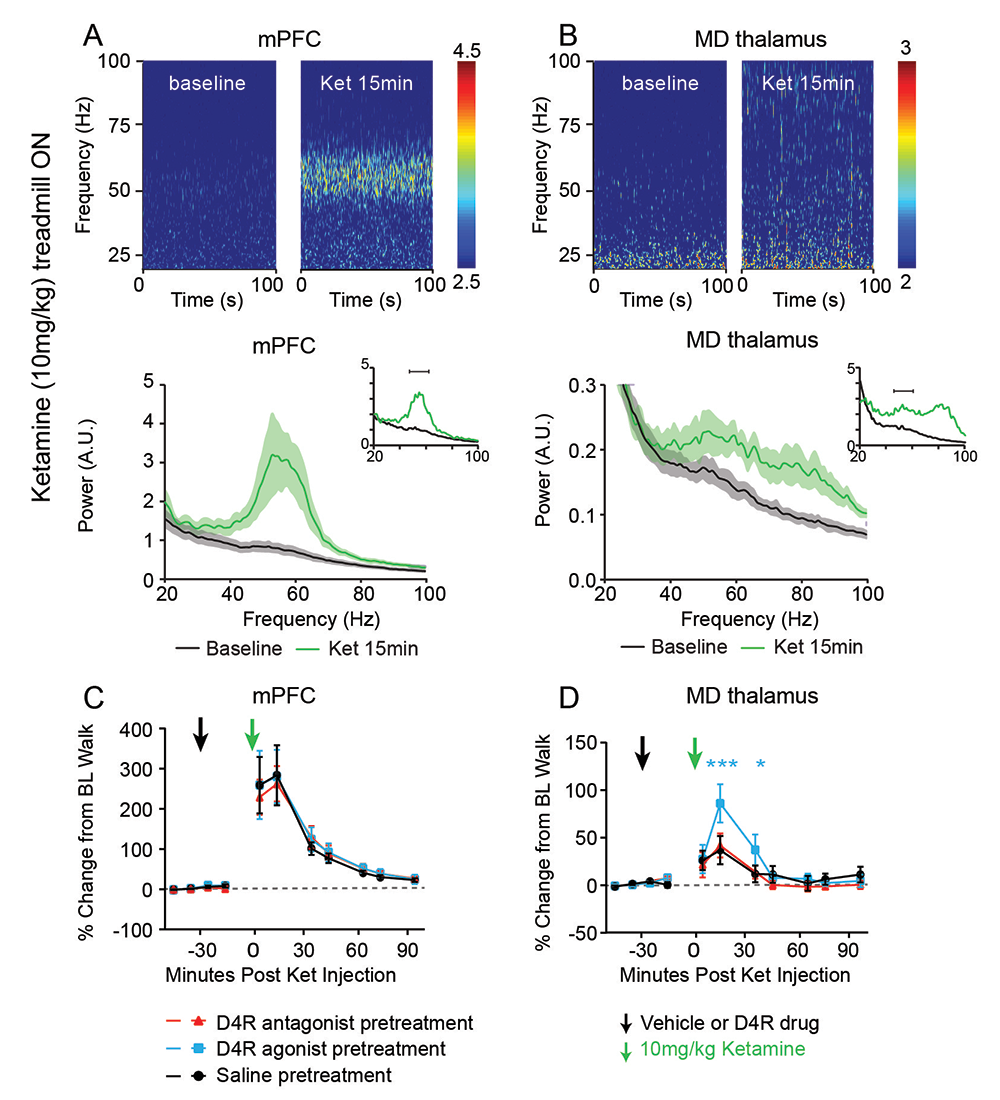
Click image to view.
Figure 5. Ketamine-induced gamma oscillations in the medial prefrontal cortex (mPFC) and medial (MD) thalamus of walking rats, and regulation by D4R agonist
Multielectrode recordings of local field potentials in the (A) mPFC and (B) MD reveal a dramatic increase in gamma frequency (40–70Hz) power in walking rats following ketamine injection (green), compared with baseline walking by rats injected with saline (black). C,D. Effects of D4R drugs on ketamine-induced gamma power. Administration of saline or a D4R antagonist (red) had no effect on ketamine-induced gamma power in either the (C) mPFC or the (D) MD thalamus, whereas the D4R agonist A-412997 (cyan) selectively increased gamma power in the MD thalamus.
Additional Funding
- Bench-to-Bedside Award (NHD15003-001-00001) “Selective Dopamine D4 Receptor-Targeting Compounds as Pro-Cognitive Drugs”
- NICHD DIR Director's Investigator Award (RRC# D-14-07). PI: Andres Buonanno; Co-PI: Juan Bonifacino. “Exploring the functional role of Neuregulin isoform diversity in CNS”
- Center for Compulsive Behaviors Fellowship Award
Publications
- Vullhorst D, Ahmad T, Karavanova I, Keating C, Buonanno A. Structural similarities between Neuregulin 1-3 isoforms determine their subcellular distribution and signaling mode in central neurons. J Neurosci 2017;37:5232-5249.
- Vullhorst D, Buonanno A. NMDA receptors regulate Neuregulin 2 binding to ER-PM junctions and ectodomain release by ADAM10. Mol Neurobiol 2019;56:8345-8363.
- Erben L, He MX, Laeremans A, Park E, Buonanno A. A novel ultrasensitive in situ hybridization approach to detect short sequences and splice variants with cellular resolution. Mol Neurobiol 2018;55:6169-6181.
- Yan L, Shamir A, Skirzewski M, Leiva-Salcedo E, Kwon OB, Karavanova I, Paredes D, Malkesman O, Bailey KR, Vullhorst D, Crawley JN, Buonanno A. Neuregulin-2 ablation results in dopamine dysregulation and severe behavioral phenotypes relevant to psychiatric disorders. Mol Psychiatry 2018;23:1233-1243.
- Skirzewski M, Karavanova I, Shamir A, Erben L, Garcia-Olivares J, Shin JH, Vullhorst D, Alvarez VA, Amara SG, Buonanno A. ErbB4 signaling in dopaminergic axonal projections increases extracellular dopamine levels and regulates spatial/working memory behaviors. Mol Psychiatry 2018;23:2227-2237.
Collaborators
- Veronica Alvarez, PhD, Section on Neuronal Structure, NIAAA, Rockville, MD
- Susan G. Amara, PhD, Laboratory of Molecular and Cellular Neurobiology, NIMH, Bethesda, MD
- Juan Bonifacino, PhD, Section on Intracellular Trafficking, NICHD, Bethesda, MD
- Jung Hwa (Susan) Cheng, PhD, EM Facility, NINDS, Bethesda, MD
- Katrina Furth, PhD, Experimental Therapeutics Branch, NINDS, Bethesda, MD
- Jennie Garcia-Olivares, PhD, Laboratory of Molecular and Cellular Neurobiology, NIMH, Bethesda, MD
- Idit Golani, MD, Technion–Israel Institute of Technology, Haifa, Israel
- Mario A. Penzo, PhD, Unit on the Neurobiology of Affective Memory, NIMH, Bethesda, MD
- Alon Shamir, PhD, Mazra Mental Health Center, Akko, Israel
- Jung-Hoon Shin, PhD, Section on Neuronal Structure, NIAAA, Rockville, MD
- Judith R. Walters, PhD, Experimental Therapeutics Branch, NINDS, Bethesda, MD
- Leqin Yan, PhD, University Medical Center Brackenridge, University of Texas, Austin, TX
Contact
For more information, email buonanno@mail.nih.gov or visit http://smn.nichd.nih.gov.


