Childhood Neurodegenerative Lysosomal Storage Disorders

- Anil B. Mukherjee, MD, PhD, Head, Section on Developmental Genetics
- Maria B. Bagh, PhD, Research Fellow
- Abhilash Appu, PhD, Visiting Fellow
- Avisek Mondal, PhD, Visiting Fellow
- Tamal Sadhukhan, PhD, Visiting Fellow
- Sondra W. Levin, MD, Adjunct Scientist
- Zhongjian (Gary) Zhang, MD, PhD, Adjunct Scientist
- Sydney Casey, AB, Postbaccalaureate Intramural Research Training Award Fellow
The Section on Developmental Genetics conducts both basic laboratory research and clinical investigations into a group of the most common childhood neurodegenerative lysosomal storage disorders (LSDs), called neuronal ceroid lipofuscinoses (NCLs), also known as Batten disease. The diseases mostly affect children and there is no effective treatment for any of the NCLs. Mutations in at least 13 different genes (called the CLNs) underlie various forms of NCLs. Among these genes, CLN1, CLN2, CLN5, CLN10, and CLN13 encode soluble lysosomal enzymes; CLN4 and CLN14 encode peripherally associated cytoplasmic proteins; CLN11 encodes progranulin, a protein in the secretory pathway; and several transmembrane proteins with various subcellular localizations are encoded by CLN3, CLN6, CLN7, CLN8, and CLN12.

Click image to view.
Section on Developmental Genetics Staff
Upper Left to Right: Sydney Casey, Abhilash Appu, Tamal Sadhukhan, and Maria Bagh.
Lower Left to Right: Sondra Levin, Gary Zhang, and Anil Mukherjee
Despite intense studies, the physiological functions of each of the CLN genes are poorly understood. Consequently, the development of mechanism-based therapeutic strategies remains challenging. Studies within the past decade have drastically changed our notion that lysosomes are merely a terminal degradative organelle. Some of the emerging new roles of the lysosome include its central role in nutrient-dependent signal transduction that regulates metabolism, cellular proliferation, or quiescence. Thus, endolysosomal dysfunction contributes to pathogenesis of virtually all LSDs.
Currently, our research focuses on understanding the molecular mechanisms of pathogenesis underlying infantile NCL (INCL: CLN1-disease), juvenile NCL (JNCL: CLN3-disease), and congenital NCL (CNCL: CLN10-disease). Interestingly, all 13 NCL types share some common clinical and pathologic features, such as intracellular accumulation of autofluorescent material, epileptic seizures, progressive psychomotor decline resulting predominantly from loss of cortical neurons in the cerebrum, neuro-inflammatory findings, visual impairment resulting from retinal degeneration, and shortened lifespan.
We first started investigating the INCL, which is caused by mutations in the CLN1 gene, a gene that encodes the lysosomal depalmitoylating enzyme palmitoyl-protein thioesterase-1 (PPT1). Numerous proteins in the body, especially in the brain, undergo posttranslational modification called S-palmitoylation (also called S-acylation), whereby a 16-carbon, saturated fatty acid (generally palmitic acid) is attached to specific cysteine residues in polypeptides via thioester linkage. While S-palmitoylation plays important roles in membrane anchorage of soluble proteins, protein-protein interaction, protein trafficking, and protein stability, these lipid-modified proteins must also be depalmitoylated for recycling or degradation and clearance by lysosomal hydrolases. PPT1 catalyzes the cleavage of thioester linkage of S-palmitoylated proteins, which is vitally important because S-palmitoylated proteins are refractory to degradation by lysosomal hydrolases. Thus, PPT1 deficiency leads to lysosomal accumulation of the S-palmitoylated proteins (constituents of ceroid), which has been proposed to be the mechanism of INCL pathogenesis. However, the precise molecular mechanism underlying INCL pathogenesis has remained elusive for more than two decades after the discovery that CLN1 mutations cause INCL. Children afflicted with INCL are normal at birth but, by 11 to 18 months of age, they exhibit signs of psychomotor retardation. By two years of age, they are completely blind owing to retinal degeneration and, by age four, they manifest no brain activity and remain in a vegetative state for several more years before eventual death. Such grim outcomes underscore the urgent need for the development of rational and effective therapeutic strategies, not only for INCL but also for all NCLs.
The aim of our clinical studies is to apply the knowledge gained from our laboratory investigations to develop novel therapeutic strategies for Batten disease. The results of our earlier investigations on INCL led to a bench-to-bedside clinical trial. Using Cln1-knockout (Cln1–/–) mice, which recapitulate virtually all clinical and pathological features of INCL, we discovered that PPT1 deficiency causes endoplasmic-reticulum (ER) and oxidative stress, which at least in part causes neuronal death by apoptosis. During the past several years, we also delineated a mechanism by which PPT1 deficiency disrupts the recycling of the synaptic vesicle (SV) proteins, which are essential for regenerating fresh SVs to replenish the SV pool size at the nerve terminals and thus maintain uninterrupted neurotransmission. We also discovered that ER and oxidative stress contribute to neuronal apoptosis and neuro-inflammation in INCL. Further, we found that PPT1 deficiency causes misrouting of the V0a1 subunit of v-ATPase (the proton pump on lysosomal membrane), which dysregulates lysosomal acidification, causing elevated pH and thus adversely affecting lysosomal degradative function.
We also developed a noninvasive method, using MRI and MRS (magnetic resonance spectroscopy), to evaluate the progression of neurodegeneration in Cln1–/– mice. The methods permit repeated evaluation of potential therapeutic agents in treated animals. Application of such methods in our clinical trial with INCL also allowed us to evaluate the progressive decline in brain volume and neurodegeneration. In collaboration with Wadih Zein, we are also conducting studies to determine whether electroretinography can be used to assess the progressive retinal deterioration in Cln1–/– as well as in the Cln1–knock-in (KI) mice generated in our laboratory, which carry the most common nonsense mutation found in the INCL patient population in the US. We also discovered that the blood-brain barrier is disrupted in Cln1–/– mice and that the pathology is ameliorated by treatment with resveratrol, which has antioxidant properties. More recently, we discovered that a nucleophilic small molecule with antioxidant properties, N-(tert-butyl)hydroxylamine (NtBuHA), ameliorates the neurological abnormalities in Cln1–/– mice and extends their lifespan. These and related studies provide insight into the complex mechanisms of heritable disorders of neurodegeneration such as INCL (CLN1 disease) and identify several potential therapeutic targets. Our results suggest that thioesterase-mimetic small molecules such as NtBuHA are potential therapeutics for INCL. More recently, we discovered that cathepsin D (CD) deficiency in lysosomes is a common pathogenic link between INCL (CLN1 disease) and congenital NCL (CNCL) or CLN10 disease. Our ongoing laboratory and clinical investigations are attempting to advance our knowledge of CLN1, CLN3, and CLN10 diseases. Our long-term plans are to apply the new findings arising from our laboratory studies to discover the pathogenic links among various NCLs and to develop novel therapeutic strategies not only for CLN1 disease but also for CLN3 and CLN10 diseases.
Palmitoyl-protein thioesterase-1 deficiency suppresses Rab7–RILP interaction dysregulating autophagy in Cln1-/- mice.
Despite the discovery more than two decades ago that PPT1 deficiency underlies INCL, the precise molecular mechanisms of pathogenesis have remained elusive. We found that autophagy is dysregulated in Cln1–/– mice, which mimic INCL, and in postmortem brain tissues from an INCL patient as well as in cultured INCL fibroblasts. Moreover, Rab7, a small GTPase, critical for autophagosome-lysosome fusion, requires S-palmitoylation for trafficking to the late endosomal/lysosomal membrane and is dysregulated in Cln1–/– mice. Notably, the defect inhibited the Rab7–RILP (Rab–interacting lysosomal protein) interaction, which is essential for Rab7 GTPase activity and required for autophagosome-lysosome fusion. The defect impaired degradative functions of the autolysosome (a hybrid structure arising from the fusion of autophagosome with lysosome), causing lysosomal accumulation of undegraded cargo, leading to INCL pathogenesis. Importantly, treatment of INCL fibroblasts with NtBuHA, a brain-penetrant, PPT1–mimetic small molecule, ameliorated the defective Rab7–RILP interaction. Our findings reveal a previously unrecognized role of CLN1/PPT1 in autophagy and suggest that thioesterase-mimetic small molecules may ameliorate the dysregulated autophagy, with therapeutic implications for INCL.
Click image to view.
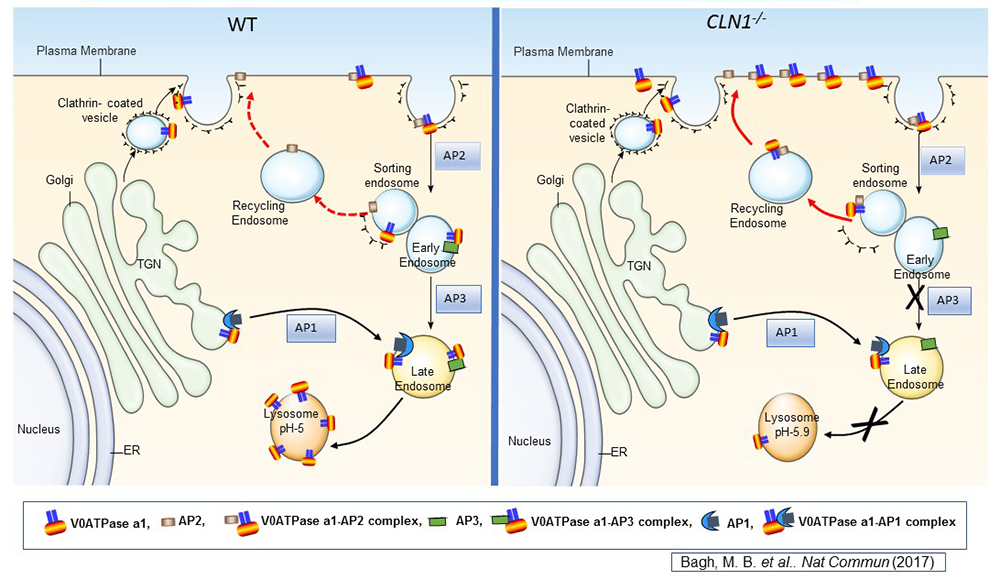
Figure 1. Lysosomal acidification defect in a mouse model of infantile neuronal ceroid lipofuscinosis
Lysosomal acidification is accomplished by vacuolar ATPase (v-ATPase) localized to the lysosomal membrane. Consisting of a lysosomal membrane–localized V0 sector and cytosolic V1 sector, v-ATPase is a multi-subunit protein. We discovered that the V0a1 subunit of the v-ATPase requires S-palmitoylation (a reversible, post-translational modification of proteins by the 16-carbon saturated fatty acid palmitate) for trafficking from the trans-Golgi network (TGN) to the lysosomal membrane. The schematic representation shows the endosomal sorting and trafficking of V0a1 in wild-type (WT) and Cln1–/– mice, which mimic INCL. In Cln1–/– mice, as opposed to WT littermates, the V0a1 subunit of v-ATPase is misrouted to the plasma membrane instead of its normal location on the lysosomal membrane. The defect dysregulates lysosomal acidification. Given that lysosomal acid hydrolases require an acidic pH in the lysosomal lumen, elevated lysosomal pH contributes to diminished degradative function of these enzymes, thereby contributing to INCL pathogenesis.
Ppt1 deficiency causes microglia proliferation and generates reactive astrocytes, mediating neuropathology in a mouse model of INCL.
In the central nervous system, the astrocytes and microglia are components of the innate immune system; however, under certain circumstances their proliferation and activation can lead to neurotoxicity. Although the role of the innate immune responses in neurodegenerative diseases have been reported to be initiated early in the disease process, the infiltration of the immune cells from the blood to the brain does not appear to play a role in the neuropathology. Recently, it was reported that the proliferation and activation of microglia leads to the conversion of astrocytes to the neurotoxic A1 phenotype, causing pathogenesis in models of common neurodegenerative diseases. In common neurodegenerative diseases such as Alzheimer’s and Parkinson’s, causal gene mutations associated with these diseases have been reported. However, establishing the cause and effect relationship linking mutations in a specific gene with neurodegeneration has remained challenging. We sought to determine whether the conversion of astrocytes to the neurotoxic A1 phenotype underlie neuropathology in a rare monogenic neurodegenerative disease such as INCL. The availability of Cln1–/– mice, a well-recognized reliable animal model of INCL, provided an opportunity to address this question. We found that in Cln1–/– mice, Ppt1 deficiency suppressed ZDHHC5 (zinc finger DHHC-type palmitoyltransferase 5) and ZDHHC23 (zinc finger DHHC-type palmitoyltransferase 23), which catalyze S-palmitoylation of the cytosolic thioesterase Apt1, which is essential for the enzyme's membrane localization. Given that Apt1 depalmitoylates membrane-localized H-Ras, reduced Apt1 levels in Cln1–/– mice significantly elevated H-Ras levels on the plasma membrane, thus activating its signaling pathway, which stimulated microglia proliferation. Increased cytokine produced by microglia and elevated complement C1q levels stimulated the generation of neurotoxic A1 astrocytes. Importantly, treating Cln1–/– mice with NtBuHA substantially ameliorated these abnormalities. Our results reveal a previously unrecognized pathway to neurodegeneration in INCL and demonstrate that NtBuHA exerts its neuro-protective effects by suppressing the generation of neurotoxic A1 astrocytes in the brain.
Click image to view.
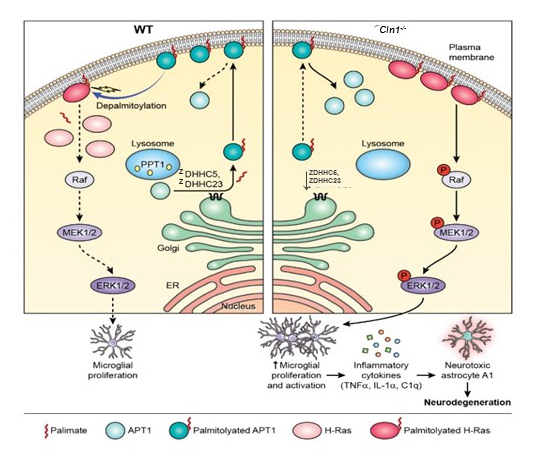
Figure 2. Dysregulated neuro-immune interaction as a mechanism of neurodegeneration in INCL
Schematic model explaining how Ppt1 deficiency in Cln1–/– mice may cause increased H-Ras signaling, which stimulates activated microglia proliferation and its conversion to A1 phenotype, leading to neurodegeneration.
Left panel: In WT mice, dynamic S-palmitoylation of APT1 mediated by ZDHHC5 and ZDHHC23 and APT1 depalmitoylation, respectively, are responsible for steady state membrane localization of both APT1 and H-Ras.
Right panel: In Cln1–/– mice, the lower level of ZDHHC5 and ZDHHC23 results in a reduced level of S-palmitoylated APT1, which increases the levels of plasma membrane–anchored, S-palmitoylated H-Ras, thus transducing proliferation signals to microglia to produce proinflammatory cytokines. The cytokines stimulate the conversion of astrocytes to the neurotoxic A1-phenotype. Elevated numbers of A1-astrocytes release yet to be identified neurotoxins, causing progressive neuronal loss in the Cln1–/– mouse brain.
Persistent mTORC1-signaling contributes to neuropathology in INCL mice: amelioration by recombinant PPT1 or Akt1 inhibitor.
Although classically, the lysosome has been considered the terminal organelle for degradation and cellular clearance, emerging evidence indicates that it also relays several nutrient cues to the master growth regulator, the mechanistic target of rapamycin complex 1 (mTORC1) kinase. Previously, we reported that in Cln1–/– mice, the reliable animal model of INCL, lysosomal acidification is dysregulated owing to mislocalization of a critical subunit (V0a1) of v-ATPase, which regulates lysosomal acidification. The v-ATPase along with Lamptor1 and SLC38A9 are essential components of the lysosomal nutrient-sensing machinery, which regulates mTORC1 signaling. We found that Lamptor1 and SLC38A9, which are components of the lysosomal nutrient-sensing apparatus, require S-palmitoylation for their localization on the lysosomal membrane and are misrouted in PPT1–deficient Cln1–/– mice. However, despite the disruption of the lysosomal nutrient-sensing machinery, mTORC1 signaling is persistently high in Cln1–/– mouse brain. Notably, in Cln1–/– mice sustained high levels of p-Akt (a serine/threonine-specific protein kinase), p-TSC2 (phospho-tubersclerosis 2), and pPRAS40 (proline-rich Akt substrate of 40 kDa), which promoted the recruitment of Rheb to the lysosomal surface, mediating mTORC1-activation. Remarkably, while mTORC1 signaling in the brain was detectable only on postnatal day 1 of wild-type (WT) mice and was virtually undetectable through adulthood, it was persistently detectable in the brain of Cln1–/– prenatal mice through adulthood. The dysregulated autophagy contributing to neuropathology. Importantly, Akt1 inhibitor or recombinant PPT1 substantially reduced mTORC1 signaling. Our results reveal a previously unrecognized role of PPT1 in regulating mTORC1 signaling and suggest that inhibitors of Akt1 may have therapeutic implications for INCL.
Misrouting of the v-ATPase subunit V0a1 dysregulates lysosomal acidification in Cln1–/– mice.
The lysosome, the primary organelle for intracellular digestion in eukaryotic organisms, contains more than 50 hydrolases, which require an acidic pH for optimal degradative function. Thus, lysosomal acidification is of fundamental importance to the degradation of macromolecules of intra- and extracellular origin that are delivered to the lysosome. Moreover, it has been reported that dysregulation of lysosomal acidification contributes to pathogenesis in virtually all LSDs, including several NCLs. Furthermore, defective regulation of lysosomal pH has also been reported in common neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease. However, despite intense studies, the mechanisms underlying the lysosomal acidification defect remain largely unclear. Lysosomal acidification is regulated by vacuolar ATPase (v-ATPase), a multisubunit protein complex composed of the cytosolic V1 sector and the lysosomal membrane–anchored V0-sector. Reversible assembly of V1/V0 sectors on the lysosomal membrane maintains functionally active v-ATPase, the proton pump of the cell.
In the mammalian genome, 23 genes encode palmitoyl-acyl-transferases (PATs), which are evolutionarily conserved, cysteine-rich zinc finger proteins containing Asp-His-His-Cys (DHHC) in the active site. Thus, the enzymes are commonly known as ZDHHC-PATs or ZDHHCs. In contrast, there are four thioesterases that have been so far characterized. Two are cytosolic (acyl-protein thioesterase-1 [Apt1] and Apt2) and two (palmitoyl-protein thioesterase-1 [PPT1] and PPT2) are localized to the lysosome. Dynamic palmitoylation (palmitoylation-depalmitoylation), requiring coordinated action of both the ZDHHC-PATs and PPTs, maintains steady-state membrane localization and the function of numerous important proteins, especially in the brain. By catalyzing depalmitoylation, thioesterases also facilitate recycling or degradation and clearance of S-palmitoylated proteins by lysosomal hydrolases.
Despite its fundamental importance, the mechanisms underlying the defect remain unclear. We found that in Cln1–/– mice, which mimic INCL, reduced v-ATPase activity correlates with elevated lysosomal pH. Moreover, v-ATPase subunit a1 of the V0 sector (V0a1) requires palmitoylation for interacting with adaptor protein-2 (AP-2) and AP-3, respectively, for trafficking to the lysosomal membrane. Notably, treatment of Cln1–/– mice with NtBuHA ameliorated this defect. Our findings reveal an unanticipated role of Cln1/Ppt1 in regulating lysosomal targeting of V0a1 and suggest that varying factors adversely affecting v-ATPase function dysregulate lysosomal acidification in other LSDs and common neurodegenerative diseases.
Cln3-mutations, which underlie juvenile NCL, cause significantly reduced levels of Ppt1 protein and Ppt1 enzyme activity in the lysosome.
Given that intracellular accumulation of ceroid is a characteristic of all NCLs, a common pathogenic link for INCL and JNCL has been suggested. It has been reported that CLN3 mutations suppress the exit of cation-independent mannose 6-phosphate receptor (CI-M6PR) from the trans-Golgi network (TGN). CI-M6PR transports soluble proteins such as PPT1 from the TGN to the lysosome, so we hypothesized that CLN3 mutations may cause lysosomal PPT1 insufficiency, contributing to JNCL pathogenesis. We found that the lysosomes in Cln3–mutant mice, which mimic JNCL, and those in cultured cells from JNCL patients, contain significantly reduced levels of Ppt1 protein and Ppt1 enzyme activity and progressively accumulate autofluorescent ceroid. Furthermore, in JNCL fibroblasts the V0a1 subunit of v-ATPase is mislocalized to the plasma membrane instead of its normal location on lysosomal membrane. The defect dysregulates lysosomal acidification, as we previously reported in Cln1–/– mice, which mimic INCL. Our findings uncover a previously unrecognized role of CLN3 in lysosomal homeostasis and suggest that CLN3 mutations causing lysosomal Ppt1 insufficiency may at least partly contribute to JNCL pathogenesis.
Publications
- Appu AP, Bagh MB, Sadhukhan T, Mondal A, Casey S, Mukherjee AB. Cln3-mutations underlying juvenile neuronal ceroid lipofuscinosis cause significantly reduced levels of palmitoyl-protein thioesterases-1 (Ppt1)-protein and Ppt1-enzyme activity in the lysosome. J Inherit Metab Dis 2019;42:944-954.
- Bagh MB, Peng S, Chandra G, Zhang Z, Singh SP, Pattabiraman N, Liu A, Mukherjee AB. Misrouting of v-ATPase subunit V0a1 dysregulates lysosomal acidification in a neurodegenerative lysosomal storage disease model. Nat Commun 2017;8:8:14612.
- Mukherjee AB, Appu AP, Sadhukhan T, Casey S, Mondal A, Zhang Z, Bagh MB. Emerging new roles of the lysosome and neuronal ceroid lipofuscinoses. Mol Neurodegener 2019;14(1):4.
Collaborators
- Eva Baker, MD, PhD, Radiology and Imaging Sciences, Clinical Center, NIH, Bethesda, MD
- Yichao Li, MS, Visual Function Core, NEI, Bethesda, MD
- Chris J. McBain, PhD, Section on Cellular and Synaptic Physiology, NICHD, Bethesda, MD
- Kenneth Pelkey, PhD, Section on Cellular and Synaptic Physiology, NICHD, Bethesda, MD
- Haohua Qian, PhD, Visual Function Core, NEI, Bethesda, MD
- Ling-Gang Wu, PhD, Synaptic Transmission Section, NINDS, Bethesda, MD
- Wadih M. Zein, MD, Ophthalmic Genetics and Visual Function Branch, NEI, Bethesda, MD
Contact
For more information, email mukherja@exchange.nih.gov or visit https://www.nichd.nih.gov/research/atNICHD/Investigators/mukherjee.


