Assembly and Function of Drosophila Chromatic Circuits

- Chi-Hon Lee, MD, PhD, Head, Section on Neuronal Connectivity
- Chun-Yuan Ting, PhD, Staff Scientist
- Yang Li, PhD, Visiting Fellow
- Jiangnan Luo, PhD, Visiting Fellow
- Pushpanathan Muthuirulan, PhD, Visiting Fellow
- Bo-mi Song, PhD, Intramural Research Training Award Fellow
- Bo Shi, MSc, Graduate Student
- Moyi Li, BA, Biological Laboratory Technician
Using the Drosophila visual system as a model, we study how neurons form complex yet stereotyped synaptic connections during development and how the assembled neural circuits extract visual attributes, such as color and motion, to guide animal behaviors. To study visual circuit functions, we combine structural and functional approaches to map visual circuits. By targeted manipulation of neuronal activity, we identified specific neurons that are functionally required for color-driven behaviors. Using both light- and electron-microscopy (EM) studies, we mapped the neuron's synaptic circuits. For circuit development, we focus on the formation of synaptic connections between the chromatic photoreceptors and their synaptic partners in the medulla neuropil. We used high-resolution imaging techniques and genetic manipulations to delineate the molecular mechanisms that control dendritic patterning and synaptic specificity of the medulla neurons.
Mapping color-vision circuits
Click image to enlarge.
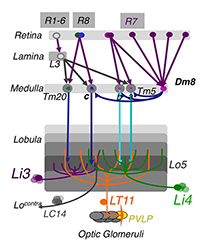
Figure 1. The chromatic circuit in Drosophila
Fly vision is mediated by three types of photoreceptors, R1–6, R7, and R8, each responding to a specific spectrum of light and connecting to different synaptic partners in the lamina and medulla neuropils. The chromatic photoreceptors R7 and R8 provide inputs for the amacrine neuron Dm8 and the transmedulla neurons Tm5a/b/c and Tm20. The transmedulla neurons transmit visual signals to four types of lobular neurons, LT11, LC14, Li3, and Li4, in the higher visual center.
Visual animals utilize spectral information in two ways: true color vision, which differentiates spectral compositions largely independently of brightness, allows animals to recognize objects and register and retrieve color memory; innate spectral preference, which is intensity-dependent and learning-independent, reflects individual species' specific ecological needs. Using a combination of genetic, histological, electrophysiological, imaging, and behavioral approaches, we study how visual circuits process chromatic information to guide behaviors in Drosophila. Our strategy is to 1) identify key neuronal types and to map their synaptic connections, 2) examine the functional requirement of identified neurons for color vision and spectral preference behaviors, and 3) determine the synaptic mechanism that transform visual signals at different processing stages.
Using molecular genetics and histology, we mapped the synaptic circuits of the chromatic photoreceptors R7 and R8 and their synaptic target neurons, the Tm and Dm neurons, in the peripheral visual system. The medulla projection (Tm) neurons (Tm5a/b/c, Tm9, and Tm20), which are analogous to vertebrate retinal ganglion cells, relay photoreceptors to higher visual centers, while the Dm (Dm8) neurons provide an indirect pathway by relaying photoreceptors to Tm neurons. To probe the synaptic connections between these identified neurons, we developed two modified versions of the GRASP (GFP reconstitution across synaptic partners) method: an activity-dependent GRASP based on split GFP–tethered synaptic proteins and a receptor-based GRASP based on split GFP–tethered neurotransmitter receptors. Using these methods, we found that the chromatic photoreceptors R7 (UV-sensing) and R8 (blue/green-sensing) provide inputs to a subset of first-order interneurons. Tm9/20/5c and Tm5a/b receive direct synaptic inputs from retinotopic R8s and R7s, respectively, consistent with their functions in processing single visual pixel information. In contrast, the amacrine neuron Dm8 pools inputs from 14 R7s and provides input for Tm5c.
To assign neurons to innate spectral preference, we systematically inactivated different first-order interneurons and examined behavioral consequences. We had previously found that the amacrine Dm8 neurons, which receive UV–sensing R7 photoreceptor inputs, are both required and sufficient for animals' innate spectral preference to UV light. We further found that inactivating Tm5c, one of Dm8's synaptic targets, abolished UV preference, indicating that Tm5c is the key downstream target for spectral preference. Both single-cell transcript profiling and immuno-histochemistry revealed that both Dm8 and Tm5c express the vesicular glutamate transporter (VGlut). RNAi knockdown of VGlut in Dm8 or Tm5c significantly reduced UV preference, suggesting the critical functions of the glutamatergic output of Dm8 and Tm5c. We further showed that Tm5c expresses four kainite-type glutamate receptors and that RNAi knockdown of these receptors, which prevents Tm5c from receiving Dm8 inputs, significantly reduced UV preference. Thus, the R7s→Dm8→Tm5c connections constitute a hard-wired glutamatergic circuit for detecting dim UV light. Two-photon calcium imaging of Dm8 and Tm5c expressing the calcium indicator GCaMP6f revealed that the activity of both neurons is suppressed by full-field UV light illumination. The light responses of Dm8 are dependent on R7's activity and Dm8's expression of the histamine chloride channel ORT, indicating that Dm8 conveys sign-inverted signal via ORT.
In contrast to UV preference, color vision in flies appears to be mediated by several partially redundant pathways. To identify color-vision circuits, we developed a novel aversive operant conditioning assay for intensity-independent color discrimination. We conditioned single flies to discriminate between equi-luminant blue or green stimuli. We found that wild-type flies can be trained to avoid either blue or green whereas mutants lacking functional R7 and R8 photoreceptors cannot, indicating that color entrainment requires the function of the narrow-spectrum photoreceptors R7s and/or R8s. Inactivating four types of first-order interneurons, Tm5a/b/c and Tm20, abolishes color learning, whereas inactivating different subsets of these neurons is insufficient to block color learning, suggesting that true color vision is mediated by parallel pathways with redundant functions. The apparent redundancy in learned color discrimination sharply contrasts with innate spectral preference, which is dominated by a single pathway, R7s→Dm8→Tm5c.
The Tm5a/b/c and Tm20 neurons relay photoreceptor signals to the higher visual center, the lobula, which in insects has been implicated in processing and relaying color information to the central brain. To determine how color information is processed in the higher visual center, we set out to identify the lobula neurons that receive direct synaptic inputs from the chromatic Tm neurons Tm5a/b/c and Tm20. We first collected and characterized 28 Gal4 lines for their expression in various lobula neurons. Second, we used our modified GRASP method to examine potential contacts between chromatic Tm neurons and the dendrites of candidate lobula neurons. We identified four types of lobula neuron that form synaptic contacts with chromatic Tm neurons: two novel lobula intrinsic neurons, Li3 and Li4, and two lobula projection neurons, LT11 and LC14. Each LT11 elaborates a large dendritic tree to cover the entire lobula layers Lo4-6 and projects an axon to optic glomeruli in the central brain. Each Li4 extends dendrites to cover about 60% of the lobula layer Lo5, while each Li3's dendrites cover about 15% of the lobular layers 5 and 6. Using the single-cell GRASP method we had developed, we further characterized the synaptic connections at the single-cell resolution and found that both Li4 and LT11 neurons receive inputs from all four chromatic Tm neurons but that LT11 appears to prefer Tm5a neurons. To confirm synaptic connections observed at the light microscopic level, we developed, for EM analysis, a two-tag EM double-labeling technique that highlights both presynaptic and postsynaptic terminals in the same preparation. By combining two orthogonal expression systems and two different peroxidases, HRP (horse radish peroxidase) and APX (ascorbate peroxidase), we expressed mitochondrion-targeted APX in Tm5c and membrane-tethered HRP in their postsynaptic neurons LT11 and confirmed appositions among synaptic profiles. Our anatomical study suggests that the lobula neurons integrate multiple chromatic inputs from Tm neurons over a large receptive field.
Dendritic development of Drosophila optic lobe neurons
Wiring brains requires routing axons and dendrites to appropriate regions, such as layers and columns, to form correct synaptic connections during development. Many neuropsychiatric disorders, such as Down syndrome, Fragile X syndrome, and Rett syndrome, have development origins and exhibit abnormal dendritic morphological defects, such as changes in branching numbers and patterns. Dendritic defects could cause neuronal connectivity defects, which likely underlie neurological and cognitive deficits. It remains unclear, however, how genetic disorders lead to dendritic patterning defects during development, resulting in erroneous connections and functional deficits in adults.
In this project, we use Drosophila optic lobe neurons as a model to study dendrite development and neural circuit assembly in the central nervous system. Like the vertebrate cortex and retina, the Drosophila optic lobe is organized in columns and layers, suggesting that the fly visual neurons and vertebrate cortex neurons face similar challenges in routing their dendrites to specific layers and columns during development. In addition, the Drosophila visual system has several unique advantages: 1) the medulla neurons extend dendritic arbors in a lattice-like structure, facilitating morphometric analysis; 2) the synaptic partnership is known; 3) genetic tools for labeling specific classes of medulla neurons and determining their connectivity are available; 4) sensitive behavioral assays are available for quantifying functional deficits. To analyze the dendritic patterns of the medulla neurons, we developed several techniques: (a) a dual-imaging technique for high-resolution imaging; (b) a registration technique to standardize and to compare dendritic patterns; (c) two modified GRASP methods for visualizing synaptic connections at the light-microscopic level; (d) a two-tag dual-labeling method for examining synapses at the electron microscopic level.
Using these techniques, we first analyzed the dendritic morphologies of four types of medulla neurons, Tm1, Tm2, Tm9 and Tm20. We identified four dendritic attributes: (i) layer-specific dendritic initiation, (ii) planar projection direction, (iii) layer-specific dendritic termination, and (iv) type-specific size of dendritic receptive field. (i) For Tm1/2/9/20, most dendritic branches (over 75%) originate from one or two primary branching nodes on the axons in the medulla M2/3 layers, while the other Tm neurons, such as Tm5a/b, initiate their dendrites in the M5/6 layers. (ii) Tm1/2/9 project their dendrites anteriorly to innervate their cognate columns while Tm20 dendrites project posteriorly. (iii) Tm1/2/9/20 project dendrites to terminate in specific layers in a type-specific fashion that matches their presynaptic partners. (iv) The dendrites of four types of Tm neurons are largely confined in single medulla columns while the amacrine neurons Dm8 extend a large dendritic tree to cover about 14 medulla columns. By clustering analyses using either PCA (principle component analysis) or an information-theory-based t-SNE (t-distributed stochastic neighboring embedding) algorithm, we found that layer-specific distribution of dendritic terminals and planar projection directions are the most important type-specific attributes and are sufficient to differentiate the Tm neurons. In sharp contrast, standard morphometric parameters, such as branch numbers and bifurcation topologies, are similar among these Tm neurons, and these parameters are thus incapable of differentiating Tm neurons dendritic patterns.
To determine the molecular mechanisms controlling dendritic patterning during development, we carried out genetic screens for morphological defects in Tm20 and Dm8 dendrites. We identified adhesion receptors, morphogen receptors, signaling molecules, and cytoskeletal regulators that are cell-autonomously required in Tm20 or Dm8 neurons for proper dendritic development. In particular, the classical cadherin N-cadherin is required in Tm20 neurons for layer-specific initiation of main dendritic branching points. Unlike wild-type Tm20 neurons, which extended most dendritic branches from one or two primary branching nodes located in the medulla M3 layer, Ncad–mutant Tm20 neurons shifted the main dendritic nodes to the M2 layers. The layer shift of the main branching nodes in Ncad–mutant Tm20 neurons correlates with an alteration of layer-specific targeting of their dendritic arbors and, to some degree, their planar projection direction. Interestingly, the total dendritic length was unaffected, suggesting that Ncad mutation specifically affects the initiation of primary dendritic branches rather than branch trimming.
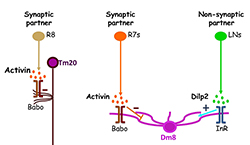
We identified two pathways that regulate the sizes of dendritic trees. We found that the TGF-beta/Activin signaling pathway negatively controls the sizes of the dendritic fields of Tm20 and Dm8. Mutant Tm20 lacking Activin signaling components, such as the receptor Baboon or the downstream transcription factor Smad2, elaborated an expanded dendritic tree, spanning several medulla columns. Morphometric analyses based on a Kaplan-Meier non-parametric estimator further showed that baboon and smad2 mutations significantly reduce dendritic termination frequency. Using a modified GRASP method, we found that the expanded dendritic tree of mutant Tm20 neurons forms aberrant synaptic contacts with several neighboring R8 photoreceptors. In contrast, wild-type Tm20 neurons form synaptic connections with single R8 photoreceptors in their cognate columns. RNAi–mediated knockdown of Activin in R7s and R8s caused an abnormal expansion of dendritic fields of Dm8 and Tm20 neurons, respectively. The results indicate that the photoreceptors R7 and R8 provide Activin specifically for their respective synaptic targets, Dm8 and Tm20. Conversely, we found that the insulin signaling pathway positively regulates the dendritic tree size of the Dm8 but not that of the Tm20 neuron. Mutant Dm8 neurons lacking insulin receptor or the downstream signaling components TOR (target of rapamycin) or Rheb (a GTPase) have a small dendritic tree while mutant Dm8 neurons lacking the negative regulators of the insulin signaling pathway, Pten (Phosphatase and tensin homolog) or TSC1 (Tuberous Sclerosis 1), have an expanded dendritic tree, as compared with the wild-type. Mutations in TOR–regulatory genes, such as TSC1/2 and AKT, have been associated with several focal malformations of cortical development (MCD) subtypes connected with epilepsy, collectively called mTORopathies. The Dm8 system we developed allows dissection of the complex phenotypes of the TOR pathway at single-cell resolution, as well as the segregation of developmental and compensatory effects.
Click image to enlarge.
Figure 2. Afferent-derived factors act in parallel pathways to regulate dendritic field sizes.
Two afferent-derived factors, Activin and Drosophila insulin-like peptide2 (Dilp2), signal on the first-order interneurons, Tm20 and Dm8, to control the sizes of their dendritic fields. Activin is provided by R7 and R8 photoreceptors, the synaptic partners of Tm20 and Dm8, respectively. Dilp2, on the other hand, is provided by lamina neurons (LNs), non-synaptic partners of Dm8. Babo, Baboon (activin receptor); InR, insulin receptor.
Additional Funding
- NIH Exploratory/Developmental Research Grant
Publications
- Li Y, Dharkar P, Han T-H, Serpe M, Lee C-H, Mayer ML. Novel functional properties of Drosophila CNS glutamate receptors. Neuron 2016;921036-1048.
- Lin T-Y, Luo J, Shinomiya K, Ting C-Y, Lu Z, Meinertzhagen IA, Lee C-H. Mapping chromatic pathways in the Drosophila visual system. J Comp Neurol 2016;524:213-227.
- Luo J, McQueen PG, Shi B, Lee C-H, Ting C-Y. Wiring dendrites in layers and columns. J Neurogenet 2016;30:69-79.
- Kulkarni A, Ertekin D, Lee C-H, Hummel T. Birth order dependent growth cone segregation determines synaptic layer identity in the Drosophila visual system. eLife 2016;5:e13715.
- Macpherson LJ, Kearney PJ, Zaharieva EE, Lin T-Y, Turan Z, Lee C-H, Gallio M. Dynamic, multiple-color labeling of neural connections by trans-synaptic fluorescence complementation. Nat Commun 2015;6:10024.
Collaborators
- Dion Dickman, PhD, University of Southern California, Los Angeles, CA
- Marco Gallio, PhD, Northwestern University, Evanston, IL
- Mary Lilly, PhD, Section on Gamete Development, NICHD, Bethesda, MD
- Mark Mayer, PhD, Laboratory of Cellular and Molecular Neurophysiology, NICHD, Bethesda, MD
- Matthew McAuliffe, PhD, Division of Biomedical Imaging Research Services Section, CIT, NIH, Bethesda, MD
- Philip McQueen, PhD, Mathematical and Statistical Computing Laboratory, CIT, NIH, Bethesda, MD
- Ian Meinertzhagen, PhD, DSc, Dalhousie University, Halifax, Canada
- Kate O'Connor-Giles, PhD, University of Wisconsin, Madison, WI
- Nishith Pandya, BA, Division of Biomedical Imaging Research Services Section, CIT, NIH, Bethesda, MD
- Thomas Pohida, MSEE, Division of Computational Bioscience, CIT, NIH, Bethesda, MD
- Randy Pursley, MSEE, Division of Computational Bioscience, CIT, NIH, Bethesda, MD
- Mihaela Serpe, PhD, Unit on Cellular Communication, NICHD, Bethesda, MD
- Mark Stopfer, PhD, Section on Sensory Coding and Neural Ensembles, NICHD, Bethesda, MD
- Benjamin White, PhD, Laboratory of Molecular Biology, NIMH, Bethesda, MD


