Thyroid Hormone Regulation of Vertebrate Postembryonic Development

- Yun-Bo Shi, PhD, Head, Section on Molecular Morphogenesis
- Liezhen Fu, PhD, Staff Scientist
- Morihiro Okada, PhD, Visiting Fellow
- Julia Rodiger, PhD, Visiting Fellow
- Yuki Shibata, PhD, Visiting Fellow
- Luan Wen, PhD, Visiting Fellow
- Nga Luu, MS, Biologist
- Lingyu Bao, MS, Graduate Student
This laboratory investigates the molecular mechanisms of thyroid hormone (TH) function during postembryonic development. The main model is the metamorphosis of Xenopus laevis and X. tropicalis, two highly related species that offer unique but complementary advantages. The control of this developmental process by TH offers a paradigm to study gene function in postembryonic organ development. During metamorphosis, different organs undergo vastly different changes. Some, like the tail, undergo complete resorption, while others, such as the limb, are developed de novo. The majority of larval organs persist through metamorphosis but are dramatically remodeled to function in a frog. For example, the tadpole intestine is a simple tubular structure consisting primarily of a single layer of larval epithelial cells. During metamorphosis, it is transformed into an organ with a multiply folded adult epithelium surrounded by elaborate connective tissue and muscles through specific larval epithelial cell death and de novo development of adult epithelial stem cells followed by their proliferation and differentiation. The wealth of knowledge from past research and the ability to manipulate amphibian metamorphosis both in vivo by using genetic approaches or hormone treatment of whole animals, and in vitro in organ cultures offer an excellent opportunity both to study the developmental function of TH receptors (TRs) and the underlying mechanisms in vivo and to identify and functionally characterize genes critical for organogenesis, particularly for the formation of the adult organ–specific stem cells, during postembryonic development in vertebrates. A major recent focus has been to make use of the TALEN and CRISPR/Cas9 technologies (References 1, 2) to knockdown or knockout the endogenous genes for functional analyses. Our past studies also revealed likely conserved mechanisms in adult intestinal stem cell development in vertebrates, which prompted us to adapt the mouse model to complement the amphibian system.
TRα controls metamorphic timing and rate but is not essential for metamorphosis.
We developed a transcription activator–like effector nuclease (TALEN) that could mutate the TRα gene in X. tropicalis with over 90% efficiency by injecting TALEN mRNAs into fertilized eggs, making it possible to analyze the role of TRα in the resulting F0 animals. Using the F0 animals, we provided evidence to support a dual function model for TRα during frog development, i.e., unliganded TRα represses gene expression and prevents precocious metamorphosis while liganded TRα activates target gene transcription and promotes metamorphosis (Reference 1). More recently, we used the F0 animals to generate animals with total knockout of the TRα gene. Surprisingly, we observed that total knockout of TRα did not block metamorphosis, suggesting that the second TR gene, TRβ, was able to mediate the metamorphic effects of TH. While TRα is not essential for metamorphosis, TRα knockout accelerated animal development, with the knockout animals reaching the onset of metamorphosis earlier. On the other hand, they were resistant to exogenous TH treatment and had slower rate of natural metamorphosis. The findings are consistent with our earlier studies with the TRα knockdown animals. Thus, our studies directly demonstrated a critical role of endogenous TRα both in mediating the metamorphic effect of TH during metamorphosis and in preventing precocious initiation of metamorphosis when TH is absent. Additionally, our knockout animal studies confirmed our initial novel observation on the growth of TRα knockdown animals, i.e., that TRα knockout enhanced tadpole growth in premetamorphic tadpoles, in part by enhancing the expression of the two growth hormone genes in Xenopus.
Click image to enlarge.
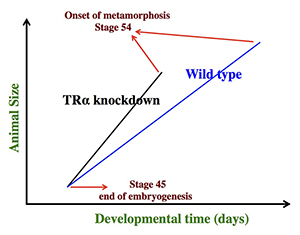
Figure 1. Schematics showing the dual effects of TRα knockdown on premetamorphic development in Xenopus tropicalis
TRα knockdown has little effect on embryogenesis, and the resulting tadpoles are normal by feeding stage (stage 45). Once feeding begins, the animals grow differently: the knockdown ones grow faster and are thus larger than wild-type siblings of the same age (in days) (compare the vertical axis values of the lines for the knockdown and wild-type animals at any given position along the horizontal axis between stages 45 and 54). The knockdown animals also have faster development, reaching developmentally more advanced stages than do wild-type siblings of the same age (in days). Thus, the knockdown animals reach stage 54, the onset of metamorphosis, at a younger age (see the horizontal axis locations for the upper end of the lines). Interestingly, when animals at stage 54 are compared, the wild-type ones are larger than the knockdown siblings, even though the latter grow faster. This is because the wild-type animals take longer to reach metamorphosis (stage 54). The extra growth time needed to reach stage 54 enables the wild-type to catch up and surpass the knockdowns in size. The results indicate that the effects of TR on growth and development are not dependent on each other (as otherwise, the size of the wild-type and knockdown animals at stage 54 would be identical).
The histone methyltransferase Dot1L is a coactivator for TR and plays an essential role in premetamorphic tadpole growth.
We earlier identified Dot1L, the only histone methyl transferase (HMT) capable of methylating histone H3K79 in vitro, as a direct target gene of TH. Interestingly, the level of H3K79 methylation is strongly enhanced by TH at TR target genes, suggesting that Dot1L is upregulated by TR and in turn functions as a TR coactivator. Indeed, in cell culture assays and in the frog oocyte transcription system, we showed that overexpression of Dot1L enhanced gene transcription by TR in the presence of TH. More importantly, transgenic overexpression of Dot1L in tadpoles also enhances gene activation of endogenous TR target genes by TH. To investigate the role of endogenous Dot1L, we generated a Dot1L–specific TALEN that was extremely efficient in mutating Dot1L when expressed in fertilized eggs of X. tropicalis, creating animals with almost no Dot1L and little H3K79 methylation. We observed that Dot1L knockdown had no apparent effect on embryogenesis (Reference 3). On the other hand, it severely retarded tadpole growth and led to tadpole lethality before metamorphosis (Reference 3). Furthermore, the expression of endogenous TR target genes was also reduced. The findings suggest that Dot1L and H3K79 methylation together play an important role in tadpole growth and development prior to metamorphosis and that Dot1L functions at least in part as a coactivator for TR.
The amidohydrolase domain–containing 1 gene is directly activated by TH and likely plays a role in the formation of adult intestinal stem cells during Xenopus metamorphosis.
TH–dependent anuran metamorphosis resembles postembryonic development in mammals, the period around birth when plasma TH levels peak. In particular, the remodeling of the intestine during metamorphosis mimics neonatal intestinal maturation in mammals when the adult intestinal epithelial self-renewing system is established. We have been using intestinal metamorphosis to investigate how organ-specific adult stem cells are formed during vertebrate development. Early studies in X. laevis showed that the process involves complete degeneration of the larval epithelium and de novo formation of adult stem cells. A tissue-specific microarray analysis of intestinal gene expression during X. laevis metamorphosis identified several candidate stem cell genes. We carried out a detailed analyses of one such gene, amidohydrolase domain–containing 1 (AMDHD1) gene, which encodes an enzyme in the histidine catabolic pathway (Reference 4). We showed that AMDHD1 is exclusively expressed in proliferating adult epithelial stem cells during metamorphosis with little expression in other intestinal tissues. We further provided evidence that TH activates AMDHD1 gene expression directly at the transcription level through TH–receptor binding to the AMDHD1 gene in the intestine. In addition, we reported earlier that the histidine ammonia-lyase (HAL) gene, another gene in histidine catabolic pathway, is similarly regulated by TH in the intestine. Taken together, the results suggest that histidine catabolism plays a critical role in the formation and/or proliferation of adult intestinal stem cells during metamorphosis.
Click image to enlarge.
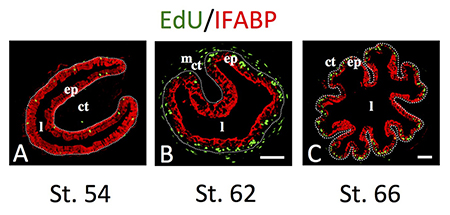
Figure 2. Intestinal metamorphosis involves the formation of clusters of proliferating, undifferentiated epithelial cells at the climax.
Tadpoles at premetamorphic stage 54 (A), climax (B, stage 62), and end of metamorphosis (C, stage 66) were injected with 5-ethynyl-2’-deoxyuridine (EdU) one hour before being sacrificed. Cross-sections of the intestine from the resulting tadpoles were double-stained by EdU labeling of newly synthesized DNA and by immunohistochemistry of IFABP (intestinal fatty acid binding protein), a marker for differentiated epithelial cells. The dotted lines depict the epithelium-mesenchyme boundary. Note that there were few EdU–labeled proliferating cells in the epithelium and that they expressed IFABP at premetamorphosis (A) and increased in the form of clustered cells (proliferating adult stem cells) that lacked IFABP at the climax of metamorphosis (B). At the end of metamorphosis, EdU–labeled proliferating cells were localized mainly in the troughs of the epithelial folds where IFABP expression was low (C). ep, epithelium; ct, connective tissue; m, muscles; l, lumen. See Reference 5 for more details.
Distinct transformations of intestinal epithelial cells during Xenopus metamorphosis revealed by molecular and cytological analyses
Earlier studies revealed several cytological and molecular markers for intestinal epithelial cells undergoing different changes during metamorphosis. However, the lack of established double labeling has made it difficult to ascertain the identities of the metamorphosing epithelial cells. We carried out double staining with several different cytological and molecular markers during TH–induced and natural metamorphosis in X. laevis (Reference 5). Our studies demonstrated conclusively that the clusters of proliferating cells in the epithelium at the climax of metamorphosis are undifferentiated epithelial cells and that they express the well known adult intestinal stem cell marker gene Lgr5. We further showed that, during metamorphosis, adult stem cells and apoptotic larval epithelial cells are distinct epithelial cells. Our findings suggest that, in response to TH during metamorphosis, morphologically identical larval epithelial cells choose two alternative paths: programmed cell death or dedifferentiation to form adult stem cells, with apoptosis occurring prior to the formation of the proliferating adult stem cell clusters (islets).
Additional Funding
- Japan Society for the Promotion of Science (JSPS) fellowships for Drs. Morihiro Okada and Yuki Shibata
Publications
- Wen L, Fu L, Guo X, Chen Y, Shi Y-B. Histone methyltransferase Dot1L plays a role in postembryonic development in Xenopus tropicalis. FASEB J 2015;29:385-393.
- Wang F, Shi Z, Cui Y, Guo X, Shi Y-B, Chen Y. Targeted gene disruption in Xenopus laevis using CRISPR/Cas9. Cell Biosci 2015;5:15.
- Wen L, Shi Y-B. Unliganded thyroid hormone receptor a controls developmental timing in Xenopus tropicalis. Endocrinology 2015;156:721–734.
- Okada M, Miller TC, Fu L, Shi Y-B. Direct activation of amidohydrolase domain-containing 1 gene by thyroid hormone implicates a role in the formation of adult intestinal stem cells during Xenopus metamorphosis. Endocrinology 2015;156:3381–3393.
- Okada M, Wen L, Miller TC, Su D, Shi Y-B. Molecular and cytological analyses reveal distinct transformations of intestinal epithelial cells during Xenopus metamorphosis. Cell Biosci 2015;5:74.
Collaborators
- Yonglong Chen, PhD, South University of Science and Technology of China, Shenzhen, China
- Atsuko Ishizuya-Oka, PhD, Nippon Medical School, Tokyo, Japan
- Daiwen Pang, PhD, Wuhan University, Wuhan, China
Contact
For more information, email shi@helix.nih.gov or visit http://smm.nichd.nih.gov.


