Regulatory Small RNAs and Small Proteins

- Gisela Storz, PhD, Head, Section on Environmental Gene Regulation
- Aixia Zhang, PhD, Staff Scientist
- Tamira K. Butler-Lively, PhD, Postdoctoral Fellow
- Michael D. Dambach, PhD, Postdoctoral Fellow
- Andrew B. Kouse, PhD, Postdoctoral Fellow
- Sahar S. Melamed, PhD, Postdoctoral Fellow
- Mona Wu Orr, PhD, Postdoctoral Fellow
- Medha V. Raina, PhD, Postdoctoral Fellow
- Taylor B. Updegrove, PhD, Postdoctoral Fellow
- Jeremy S. Weaver, PhD, Postdoctoral Fellow
- Joshua M. Mills, BS, Postbaccalaureate Fellow
- Aracely A. Romero, BS, Postbaccalaureate Fellow
- Hira Shabbir, BS, Postbaccalaureate Fellow
- Hanbo Wang, BS, Graduate Student
Currently, we have two main interests: identification and characterization of small noncoding RNAs and identification and characterization of small proteins of less than 50 amino acids. Both small RNAs and small proteins have been overlooked because they are not detected in biochemical assays and the corresponding genes are poorly annotated and missed in genetic screens. However, mounting evidence suggests that both classes of small molecules play important regulatory roles.
Identification and characterization of small regulatory RNAs
During the past 15 years, we have carried out several different systematic screens for small regulatory RNA (sRNA) genes in Escherichia coli. The screens included computational searches for conservation of intergenic regions and direct detection after size selection or co-immunoprecipitation with the RNA–binding protein Hfq. We recently examined sRNA expression using deep sequencing to extend our identification of sRNAs, particularly antisense RNAs (Thomason MK et al. J Bacteriol 2015;197:18).
A major focus for the group has been to elucidate the functions of the sRNAs we and others identified. Early on, we showed that the OxyS RNA, whose expression is induced in response to oxidative stress, acts to repress translation through limited base pairing with target mRNAs. We discovered that OxyS action is dependent on the Sm-like Hfq protein, which acts as a chaperone to facilitate OxyS RNA base pairing with its target mRNAs (Gottesman S, Storz G. RNA 2015;21;511). Recently, we carried out extensive mutational studies of Hfq (Shu DJ et al. EMBO J 2015;34:2557). The analysis revealed that amino acids on three different RNA–interaction surfaces—the proximal face, the distal face, and the rim of the doughnut-shaped protein—differentially impact Hfq association with sRNAs and their mRNA targets (Reference 1) (Figure 1).
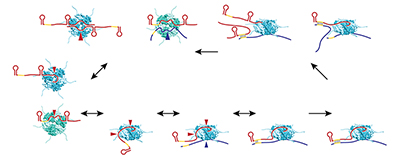
Click image to enlarge.
Figure 1. Model of Hfq–facilitated base pairing between sRNAs and mRNAs
E. coli Hfq (teal for the proximal face and rim views and green for the distal face view) employs four solvent-exposed surfaces to interact with RNA; sRNAs (red) have been found to contact the proximal and distal faces, rim, and C-terminus, and mRNAs (blue) have been shown to contact the distal face, rim, and C-terminus. Red and blue arrows denote sRNA and mRNA binding to Hfq, respectively. Hfq binding to mRNAs and sRNAs is thought to occur in random order. Initial binding is likely to involve only a subset of subunits, allowing for rapid displacement by other RNAs. Many RNAs bind to multiple surfaces, resulting in changes in RNA secondary structure and protection against RNase degradation, particularly for sRNAs. The free surface(s) of Hfq not already bound to RNA interacts with the cognate RNA partner, positioning the unbound seed region of the sRNA in close proximity to the unbound complementary region of the mRNA, facilitating base pairing. The lower affinity of duplex RNA for Hfq causes free RNAs to compete off sRNA–mRNA pairs, allowing Hfq to serve as a matchmaker for another pair of RNAs.
It is now clear that Hfq–binding sRNAs, which act through limited base pairing, are integral to many different stress responses in E. coli and other bacteria (Reference 2). For example, we showed that the Spot 42 RNA, whose levels are highest when glucose is present, plays a broad role in catabolite repression by directly repressing genes involved in central and secondary metabolism, redox balancing, and the consumption of diverse nonpreferred carbon sources. Similarly, we discovered that a Sigma(E)–dependent sRNA, MicL, transcribed from a promoter located within the coding sequence of the cutC gene, represses synthesis of the lipoprotein Lpp, the most abundant protein in the cell, to oppose membrane stress. We found that the copper-sensitivity phenotype previously ascribed to inactivation of the cutC gene is actually derived from the loss of MicL and elevated Lpp levels. The observation raises the possibility that other phenotypes currently attributed to protein defects are attributable to deficiencies in unappreciated regulatory RNAs (Reference 3). Most recently, we characterized a set of sRNAs expressed from a locus we denoted sdsN (Reference 4). Two longer sRNAs, SdsN137 and SdsN178, are transcribed from two Sigma(S)–dependent promoters but share the same terminator. Whole-genome expression analysis after pulse overexpression of SdsN137 and assays of lacZ fusions revealed that the SdsN137 directly represses the synthesis of the nitroreductase NfsA, which catalyzes the reduction of the nitrogroup (NO2) in nitroaromatic compounds and the flavohemoglobin HmpA, which has aerobic nitric oxide (NO) dioxygenase activity. Consistent with this regulation, SdsN137 confers resistance to nitrofurans. Interestingly, SdsN178 is defective in regulating the above targets owing to unusual binding to the Hfq protein, but cleavage leads to a shorter form, SdsN124, which is able to repress nfsA and hmpA.
In addition to sRNAs that act via limited base pairing, we have been interested regulatory RNAs that act by other mechanisms. For example, early work showed that the 6S RNA binds to and modulates RNA polymerase by mimicking the structure of an open promoter. In a more recent study, we discovered that a broadly conserved RNA structure motif, the yybP-ykoY motif, found in the 5′-UTR of the mntP gene encoding a manganese exporter, directly binds to manganese, resulting in a conformation that liberates the ribosome-binding site (Reference 5). Remarkably, we were able to recapitulate the effect of manganese-dependent activation of translation in vitro. We also found that the yybP-ykoY motif responds directly to manganese ions in Bacillus subtilis. The identification of the yybP-ykoY motif as a manganese ion sensor points to the genes that are preceded by this motif, encode a diverse set of poorly characterized membrane proteins, and play roles in metal homeostasis.
Studies are ongoing to further characterize other Hfq–binding RNAs and their evolution as well as antisense RNAs and sRNAs that act in ways other than base pairing.
Identification and characterization of small proteins
In our genome-wide screens for sRNAs, we found that several short RNAs do encode small proteins. The correct annotation of the smallest proteins is one of the biggest challenges of genome annotation, and perhaps more importantly, few annotated short open reading frames (ORFs) have been documented to correspond to synthesized proteins. Although these proteins have largely been missed, the few small proteins that have been studied in detail in bacterial and mammalian cells were shown to have important functions in signaling and in cellular defenses (Reference 6). We thus established a project to identify and characterize proteins of less than 50 amino acids.
We used sequence conservation and ribosome binding-site models to predict genes encoding small proteins, defined as having 16–50 amino acids, in the intergenic regions of the model E. coli genome. We tested expression of the predicted as well as previously annotated small proteins by integrating the sequential peptide affinity tag directly upstream of the stop codon on the chromosome and assaying for synthesis using immunoblot assays. The approach confirmed that 20 previously annotated and 18 newly discovered proteins of 16–50 amino acids are synthesized. We are now establishing complementary biochemical approaches to identify additional small proteins.
Remarkably more than half the newly discovered proteins are predicted to be single transmembrane proteins, an observation that prompted us to examine the localization, topology, and membrane insertion of the small proteins. Biochemical fractionation showed that, consistent with the predicted transmembrane helix, the small proteins are generally most abundant in the inner membrane fraction. Examples of both Nin-Cout and Nout-Cin orientations as well as dual topology were found in assays of topology-reporter fusions to representative small transmembrane proteins. In addition, fractionation analysis of small-protein localization in mutant strains uncovered differential requirements for these membrane-insertion pathways. Thus, despite their diminutive size, small proteins display considerable diversity in topology, biochemical features, and insertion pathways.
We now are employing many of the approaches the group has used to characterize the functions of sRNAs to elucidate the functions of the small proteins. Systematic assays for the accumulation of tagged versions of the proteins showed that many small proteins accumulate under specific growth conditions or after exposure to stress. We also generated and screened bar-coded null mutants and identified small proteins required for resistance to cell envelope stress and acid shock. In addition, various tagged derivatives are being exploited to identify co-purifying complexes. The combination of these approaches is giving insights into when, where, and how the small proteins are acting.
For example, we found that synthesis of the 42–amino acid protein MntS is repressed by high levels of manganese through the manganese transporter regulator MntR. We showed that MntS helps manganese activate a variety of enzymes under manganese-poor conditions, while overproduction of MntS leads to very high intracellular manganese and bacteriostasis under manganese-rich conditions (Reference 7). These and other phenotypes led us to propose that MntS modulates intracellular manganese levels, possibly by inhibiting the manganese exporter MntP.
We also discovered that the 49-amino acid protein AcrZ associates with the AcrAB-TolC multidrug efflux pump, which confers resistance to a wide variety of antibiotics and other compounds. Co-purification of AcrZ with AcrB, in the absence of both AcrA and TolC, two-hybrid assays, and suppressor mutations indicate that this interaction occurs through the inner membrane protein AcrB. Mutants lacking AcrZ are sensitive to many, but not all, antibiotics transported by AcrAB-TolC. The differential antibiotic sensitivity suggests that AcrZ may enhance the ability of the AcrAB-TolC pump to export certain classes of substrate. This work, together with our ongoing studies on other small proteins, suggests that many are acting as regulators of larger membrane proteins.
Publications
- Updegrove TB, Zhang A, Storz G. Hfq: the flexible RNA matchmaker. Curr Opin Microbiol 2016;30:133-138.
- Machner MP, Storz G. Infection biology: small RNA with a large impact. Nature 2016;529:472-473.
- Updegrove TB, Shabalina SA, Storz G. How do base-pairing small RNAs evolve? FEMS Microbiol Rev 2015;39:379-391.
- Hao Y, Updegrove TB, Livingston NN, Storz G. Protection against deleterious nitrogen compounds: role of sigmaS-dependent small RNAs encoded adjacent to sdiA. Nucleic Acids Res 2016;44:6935-6948.
- Dambach M, Sandoval M, Updegrove TB, Anantharaman V, Aravind L, Waters LS, Storz G. The ubiquitous yybP-ykoY riboswitch is a manganese-responsive regulatory element. Mol Cell 2015;57:1099-1109.
- Storz G. New perspectives: insights into oxidative stress from bacterial studies. Arch Biochem Biophys 2016;595:25-27.
- Martin JE, Waters LS, Storz G, Imlay JA. The Escherichia coli small protein MntS and exporter MntP optimize the intracellular concentration of manganese. PLoS Genet 2015;11:e1004977.
Collaborators
- Susan Gottesman, PhD, Laboratory of Molecular Biology, NCI, Bethesda, MD
- James A. Imlay, PhD, University of Illinois, Urbana, IL
- Aravind L. Iyer, PhD, National Center for Biotechnology Information, NIH, Bethesda, MD
- Kay Nieselt, PhD, Universität Tübingen, Tübingen, Germany
- Svetlana A. Shabalina, PhD, National Center for Biotechnology Information, NIH, Bethesda, MD
- Cynthia M. Sharma, PhD, Research Centre for Infectious Diseases, Universität Würzburg, Germany
- Lauren S. Waters, PhD, University of Wisconsin, Oshkosh, WI
Contact
For more information, email storz@helix.nih.gov or visit http://storz.nichd.nih.gov.


