Quantitative Imaging and Tissue Sciences

- Peter J. Basser, PhD, Head, Section on Quantitative Imaging and Tissue Sciences
- Ferenc Horkay, PhD, Staff Scientist
- Carlo M. Pierpaoli, MD, PhD, Staff Scientist/NIBIB Stadtman Tenure-Track Investigator
- Ruiliang Bai, PhD, Visiting Fellow
- Dan Benjamini, PhD, Visiting Fellow
- Beatriz P. Betancourt, PhD, Postdoctoral Intramural Research Training Award Fellow
- Nathan Hu Williamson, PhD, Postdoctoral Intramural Research Training Award Fellow
- Neda Sadeghi, PhD, Postdoctoral Intramural Research Training Award Fellow
- Adam Bernstein, MD, Predoctoral Intramural Research Training Award Fellow
- Amber Simmons, BS, Postbaccalaureate Intramural Research Training Award Fellow
- Elizabeth Hutchinson, PhD, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
- Okan Irfanoglu, PhD, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
- Jeffrey Jenkins, MS, Contractor funded by Congressionally Directed Medical Research Programs (CDMRP) and Catholic University of America
- Sarah King, PhD, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
- Michal Komlosh, PhD, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
- Amritha Nayak, MS, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
- Laura Reyes, PhD, Contractor funded by the Henry Jackson Foundation-Center for Neuroscience and Regenerative Medicine
In our tissue sciences research, we strive to understand fundamental relationships between function and structure in living tissues, using ‘engineered’ tissue constructs and tissue analogs. Specifically, we are interested in how microstructure, hierarchical organization, composition, and material properties of tissues affect their biological function or dysfunction. We investigate biological and physical model systems at various length and time scales, performing physical measurements in tandem with developing physical/mathematical models to explain their functional properties and behavior. Experimentally, we use water to probe both equilibrium and dynamic interactions among tissue constituents from nanometers to centimeters and from microseconds to lifetimes. To determine the equilibrium osmo-mechanical properties of well defined model systems, we vary water content or ionic composition systematically. To probe tissue structure and dynamics, we employ atomic force microscopy (AFM), small-angle X-ray scattering (SAXS), small-angle neutron scattering (SANS), static light scattering (SLS), dynamic light scattering (DLS), and one- and two-dimensional nuclear magnetic resonance (NMR) relaxometry and diffusometry. A goal of our basic tissue sciences research is to develop tools that can be translated from bench-based quantitative methodologies to the bedside.
Our tissue sciences activities dovetail with our basic and applied research in quantitative imaging that is intended to generate measurements and maps of intrinsic physical quantities, including diffusivities, relaxivities, or exchange rates, rather than qualitative stains and images conventionally used in neuro-radiology. Our quantitative imaging group uses knowledge of physics, engineering, applied mathematics, imaging and computer sciences, and insights gleaned from our tissue sciences research to discover and develop novel imaging biomarkers that sensitively and specifically detect changes in tissue composition, microstructure, or microdynamics. The ultimate translational goal of developing such biomarkers is to assess normal and abnormal development, diagnose childhood diseases and disorders, and characterize degeneration and trauma. Primarily, we use MRI as our imaging modality of choice because it is well suited for many NICHD mission–critical applications; it is non-invasive, non-ionizing, requires in most cases no exogenous contrast agents or dyes, and is deemed safe for use with fetuses and children in both clinical and research settings.
A technical objective has been to transform clinical MRI scanners into scientific instruments capable of producing reproducible, highly accurate, and precise imaging data to measure and map useful imaging quantities for various applications, including single scans, longitudinal and multi-site studies, personalized medicine, genotype/phenotype studies, and for populating imaging databases with high-quality normative data.
In vivo MRI histology
We aim to develop novel next-generation in vivo MRI methods to better understand brain structure and function in normal and abnormal development, disease, degeneration, and trauma. The most mature technology that we invented, developed, and translated is Diffusion Tensor MRI (DTI), by which we measure D, a diffusion tensor of water, voxel-by-voxel within an imaging volume. Information derived from this quantity includes white-matter fiber-tract orientation, the mean-squared distance that water molecules diffuse in each direction, the orientationally averaged mean diffusivity, and other intrinsic scalar (invariant) quantities. These imaging parameters behave like non-invasive quantitative histological ‘stains’ obtained by probing endogenous tissue water in vivo without requiring exogenous contrast agents or dyes. The bulk or orientationally averaged apparent diffusion coefficient (mean ADC) is the most successful and widely used DTI parameter to identify ischemic regions in the brain during acute stroke and to follow response to therapy in tumors. Our measures of diffusion anisotropy (e.g., fractional anisotropy or FA) are universally used to follow changes in normally and abnormally developing white matter, including dysmyelination and demyelination. Our group also pioneered the use of fiber direction–encoded color (DEC) maps to display the orientation of the main association, projection, and commissural white matter pathways in the brain. To assess anatomical connectivity among various cortical and deep brain gray matter areas, we also proposed and developed DTI “Streamline” Tractography.
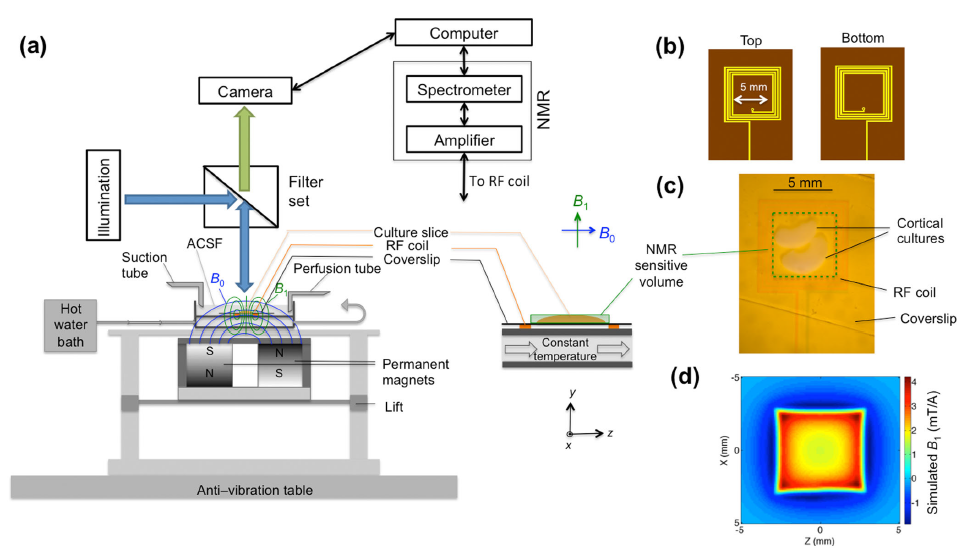
Click image to enlarge.
Figure 1. Novel instrument for simultaneous functional MR and calcium imaging studies
Schematic diagram of a novel instrument for simultaneous functional MR and calcium imaging studies of organotypic cultured brain tissue. (a) Schematic diagram of the simultaneous MR and fluorescence imaging test bed (left) and an enlargement of the components near the organotypic cultured tissue (right), which is immersed in artificial cerebral spinal fluid (ACSF). (b) Top and bottom layers of the two-layer multi-turn radio-frequency (RF) surface coil. (c) Real image of the coil with the cortical culture mounted under approximately 0.63Å magnification. (d) Simulated two-dimensional B1 field distribution at y = 0.2 mm in the x–z plane. This device is allowing us to test existing fMRI methods without cardiovascular, respiratory, or other artifacts and confounds. It also provided a test-bed for us to develop new fMRI approaches.
More recently, we invented and developed a family of advanced in vivo diffusion MR methods to measure fine-scale microstructural features of axons and fascicles, which otherwise could only be measured using laborious ex vivo histological methods. We have been developing efficient means for performing “k and q-space MRI” in the living brain, such as “Mean Apparent Propagator” (MAP) MRI. This approach detects subtle microstructural and architectural features in both gray and white matter at micron-scale resolution, several orders of magnitude smaller than the MRI voxel. It also subsumes DTI, as well as providing a bevy of new in vivo quantitative ‘stains’ to measure and map. We also developed a family of diffusion MRI methods to ‘drill down into the voxel’ and measure features such as average axon diameter (AAD) and axon diameter distribution (ADD) within large white-matter fascicles, dubbing them CHARMED and AxCaliber MRI, respectively. After careful validation studies, we reported the first in vivo measurement of ADDs within the rodent corpus callosum. The ADD is functionally important given that axon diameter helps determine conduction velocity and therefore the rate at which information flows along white matter pathways as well as delays or latencies between and among different brain areas. We then developed a companion mathematical theory to explain the observed ADDs in different fascicles, suggesting that they represent a trade-off between information flow and metabolic demands. We also developed novel multiple pulsed-field gradient (mPFG) methods and demonstrated their feasibility for use in vivo on conventional clinical MRI scanners as a further means to extract quantitative features to measure and map in the central nervous system (CNS). The methods can also provide an independent measurement of the AAD and other features of cell size and shape.
Although brain gray matter appears featureless in DTI maps, its microstructure and architecture are rich and varied throughout the brain, not only along the brain's cortical surface, but also within and among its various cortical layers and within deep gray matter regions. To target this tissue, we have been developing several noninvasive, in vivo methods to measure unique features of cortical gray matter microstructure and architecture that are currently invisible in conventional MRI. One goal is to ‘parcellate’ or segment the cerebral cortex in vivo into its approximately 500 distinct cyto-architechtonic areas. To this end, we are developing advanced MRI sequences to probe correlations among microscopic displacements of water molecules in the neuropil as well as sophisticated mathematical models to infer distinguishing microstructural and morphological features of gray matter. Within the past year, we pioneered and developed several promising two-dimensional MRI relaxometry, exchange, and diffusometry methods, which we plan to use to study water mobility and exchange in gray matter. We believe these will have promise in identifying inflammation and redistribution of tissue water in brain parenchyma.
In general, we are continuing to develop translationally oriented methods to follow normal and abnormal development, aid in the diagnosis of various diseases and disorders of the brain, noninvasively and in vivo, and provide information to help neurosurgeons plan operations and interventions.
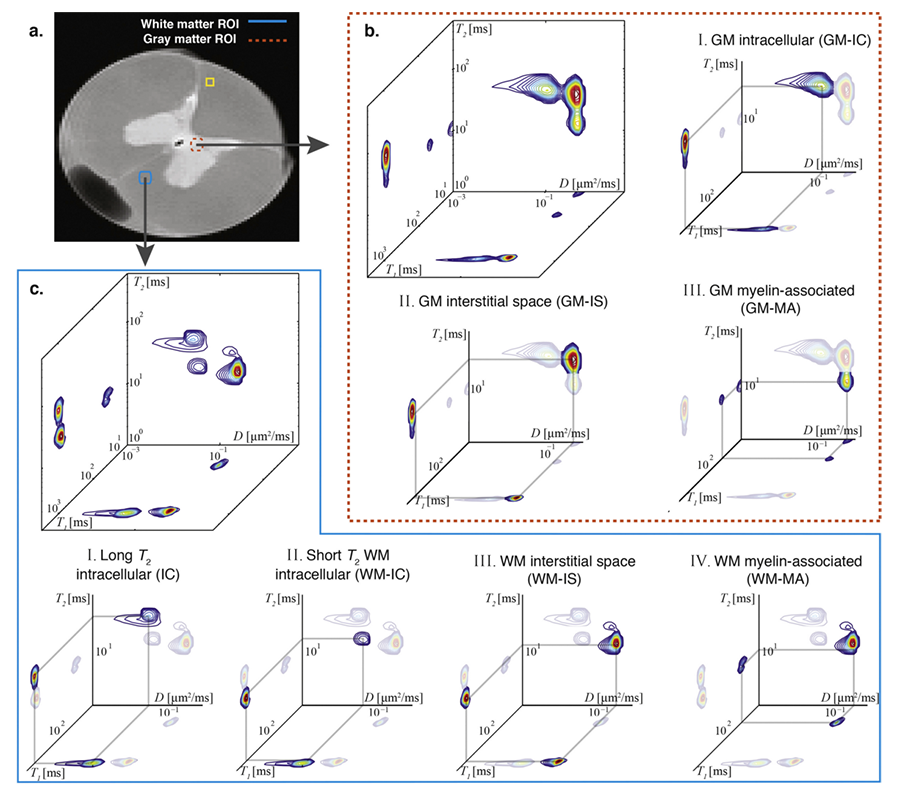
Click image to enlarge.
Figure 2. Two-dimensional relaxometry MRI of ferret spinal cord
Spectra from gray matter (GM) and white matter (WM) regions of interest (ROIs). (a) A high-resolution proton density MR image is shown with the locations of representative ROIs: WM (blue box) and GM (red box) ROIs. After voxel-wise analysis, the spectra in each of the GM and WM ROIs were averaged and are presented in (b) and (c), respectively. The different peaks are assigned to microscopic neuro-anatomical components according to their respective diffusion (D), T1, and T2 values. Assignments of different spectra to cellular and tissue components can be made using histological images obtained from the same slice. GM: I. Intracellular (GM-IC); II. GM interstitial space (GM-IS); and III. GM myelin-associated (GM-MA). WM: I. Long T2 intracellular (IC); II. Short T2 intracellular (WM-IC); III. WM interstitial space (WM-IS); and IV. WM myelin-associated (WM-MA).
Quantitative pediatric MRI
MRI is considered safer than X-ray–based methods, such as computed tomography (CT), for scanning infants and children. However, clinical MRI data still lacks the quantitative character of CT data. Clinical MRI relies upon the acquisition of ‘weighted images,’ whose contrast is affected by many factors, some intrinsic to the tissue and some dependent on the details of the experiment and experimental design. The diagnostic utility of conventional MRI for many neurological disorders is unquestionable. However, the scope of conventional MRI applications is limited to revealing either gross morphological features or focal abnormalities, which result in regional differences in signal intensities within a given tissue. To detect pathology, conventional MRI relies on differences in contrast between areas that are presumed ‘affected’ and those presumed ‘normal,’ rendering it intrinsically insensitive to subtle global changes that may affect the entire tissue or organ. Clinical MRI also lacks biological specificity. Although quantification per se does not ensure improved specificity, it is nonetheless necessary for developing robust and reliable imaging ‘biomarkers.’ In particular, MRI assessment of normal brain development and developmental disorders has benefiting greatly from the introduction of ‘quantitative’ clinical MRI techniques, in which one obtains maps of meaningful physical quantities or chemical variables that can be measured in physical units and compared among different tissue regions, in both longitudinal and cross-sectional studies. Quantitative MRI methods, such as DTI, also increase sensitivity, providing a basis for monitoring subtle changes that occur, e.g., during the progression or remission of disease, by comparing measurements in a single subject with normative values acquired in a healthy population. Quantitative MRI methods should also aid in precision imaging studies, whereby MRI phenotypic data can be meaningfully linked to a subject's genotype.
Our group has been carrying out several clinical studies that utilize novel quantitative MRI acquisition and analysis methods and whose aim is to improve accuracy and reproducibility in diagnosis and to detect and follow normal and abnormal development. These studies include the following:
- The NIH Study of Normal Brain Development, jointly sponsored by a consortium of four NIH Institutes (NICHD, NIMH, NINDS, and NIDA), was a multi-center effort to advance our understanding of normal brain development in typical healthy children and adolescents. The Brain Development Cooperative Group (http://www.brain-child.org), created by this mechanism, is still active, publishing numerous papers each year, primarily by mining these rich data. Tandem structural MRIs and standardized neuropsychological tests performed on this population are also available to researchers outside the consortium. Our role in this interdisciplinary project was as the DTI Data-Processing Center (DPC). While we have now processed all admissible DTI data and uploaded them to a database accessible to all interested investigators, we continue to mine and analyze the data, having recently developed age-specific DTI atlases of normal brain development. We also publicly released various versions of our software package, TORTOISE, which we developed for this project (and related documentation), which can be downloaded from http://www.tortoisedti.org. We continue to support and update the software. We are continuing to use this advanced DTI–processing pipeline to produce high-quality normative data from the project, which we make publicly available through the National Database for Autism Research (NDAR; https://nda.nih.gov).
- In collaboration with Susan Swedo, we studied autistic subjects using DTI and quantitative MRI relaxometry methods. While several MRI studies reported abnormal features in the autistic brain, no clear MRI ‘biomarker’ of autism exists. The aim of the study is to use robust quantitative metrics to identify potential anatomical abnormalities in the autistic brain and to find candidate imaging biomarkers for this disorder.
- In collaboration with Katherine Warren, we acquired quantitative MRI data in children with pontine gliomas to identify MRI prognostic factors. With John Park we scanned subjects with supratentorial gliomas to distinguish recurrence from radiation necrosis.
- In collaboration with Filippo Arrigoni, we use multi-modal MR imaging (DTI, fMRI, and quantitative relaxometry) to evaluate cerebral reorganization caused by various rehabilitation protocols in children with cerebral palsy and traumatic brain injury (TBI). We collected diffusion MRI data on subjects affected by the pure form of hereditary spastic paraparesis, as well as those with additional cognitive impairment. There are remarkable neuro-anatomic differences between the two groups.
- We are continuing to work under the auspices of the Center for Neuroscience and Regenerative Medicine (CNRM), DoD, to investigate potential plasticity changes after rehabilitation in military personnel affected by TBI or post-traumatic stress disorder (PTSD). We are also exploring the potential clinical utility of MAP-MRI to provide quantitative imaging biomarkers that are more sensitive and specific than those provided by DTI, to determine whether they could be used to detect and assess mild TBI (mTBI).
- We received grant support from the Congressionally Directed Medical Research Program (CDMRP) to investigate, in collaboration with Sharon Juliano, a ferret model of mTBI, using advanced MRI methods, particularly MAP-MRI, combined with histopathological techniques provided by Juliano's laboratory. We are currently acquiring MRI data.
- In collaboration with the Veteran’s Administration, DoD, and the Chronic Effects of Neurotrauma Consortium (CENC), we are enabling the acquisition of multi-site DTI data from various clinical centers in the VA network.
Our involvement in TBI research, particularly in detecting mTBI, has continued to expand, because it is of high relevance to the NICHD mission, it is an acute problem in the pediatric population, and it afflicts men and women in the military. DTI provides essential information for the diagnosis of TBI and has the potential to be developed into an important tool for the assessment of potential structural damage in PTSD. For clinical applications, however, reliable imaging protocols are needed. Part of our work is to develop a robust DTI data–processing pipeline in order to improve the accuracy and reproducibility of DTI findings for CNRM investigators and for the larger clinical and scientific community involved in TBI research. To this end, we are adding new modules to our existing state-of-the-art DTI data-processing pipeline as well as tools to permit calibration of DTI experiments, using our novel polyvinyl pyrrolidone (PVP) polymer–based diffusion MRI phantom that we developed, patented, and disseminating to a number of clinical sites.
Looking ahead, to permit analysis of novel MRI data such as those described above, as well as to develop new clinical and biological applications of quantitative MRI, we need to create a mathematical, statistical, and image sciences–based infrastructure. To date, we have developed algorithms that generate a continuous, smooth approximation of the discrete, noisy, measured DTI field data so as to reduce noise and allow us to follow fiber tracts more reliably. We proposed a novel Gaussian distribution for tensor-valued random variables that we used in designing optimal DTI experiments and interpreting their results. In tandem, we developed non-parametric empirical (e.g., Bootstrap) methods to determine the statistical distribution of DTI–derived quantities in order to study, for example, the inherent variability and reliability of computed white-matter fiber-tract trajectories. Such parametric and non-parametric statistical methods enable us to apply powerful hypothesis tests to assess the statistical significance of findings in a wide range of important biological and clinical applications that are currently being tested using ad hoc statistical methods. We are also developing novel methods to register or warp different brain volumes and to generate group-average data or atlases from various subject populations. Recently, our group has been developing methods for studying the reproducibility and reliability of different tractography methods, given their widespread use to assess anatomical connections between different brain regions in vivo. In the area of artifact remediation and correction, we pioneered methods to correct for subject motion and for artifacts caused by induced eddy-current and echo-planar imaging (EPI) distortion. However, much work remains to be done in order to address and remedy MRI artifacts to permit one to draw statistically significant inferences from clinical DTI data, obtained in longitudinal and multi-center studies, particularly single-subject studies.
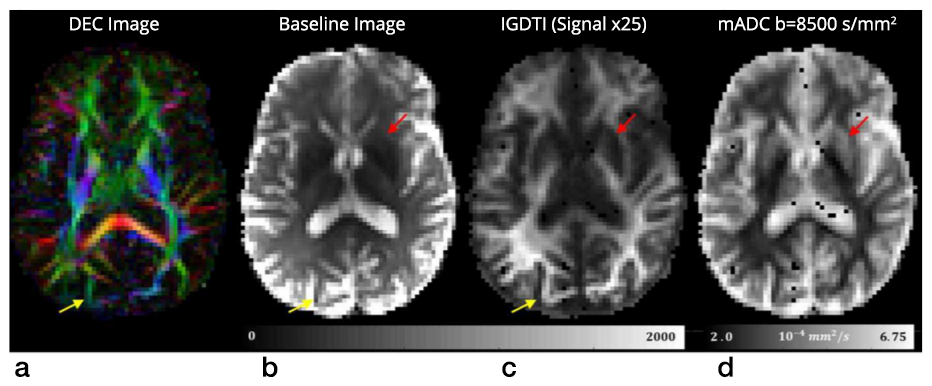
Click image to enlarge.
Figure 3. New rapid isotropically weighted MRI method for fetal and pediatric MRI applications
Isotropically weighted generalized diffusion tensor imaging (IGDTI) provides a rapid means of obtaining diffusion MRI data that is intrinsic and insensitive to diffusion anisotropy. We proposed a new experimental design and analysis framework to measure orientationally averaged diffusion-weighted images (DWIs), which removes the modulation in image intensity attributable to anisotropic structures, such as brain white matter. The approach is expected to have significant applications in neuro-radiology, particularly for fetal and pediatric imaging, where subject movement can be a significant confound.
Biopolymer physics: water-ion-biopolymer interactions
A major focus of this basic research project is to understand the effect of ion-water-biopolymer interactions. These are ubiquitous in biology, ranging from the movement of water and ions across channels, to the self-assembly of aberrant proteins into nanofibers in neuro-degenerative processes, such as Creutzfeldt-Jakob disease, Alzheimer’s disease, and chronic traumatic encephalopathy (CTE). Despite their importance and prevalence, however, little is understood about these interactions from a physics perspective.
To address this dearth in our understanding, we developed a multi-scale experimental framework to study these interactions by combining macroscopic techniques (e.g., osmotic swelling pressure measurements, mechanical measurements) with higher-resolution scattering methods (e.g., SANS and SAXS). Swelling pressure measurements provide information on the overall thermodynamic response, while SANS and SAXS allow us to investigate biopolymers at the molecular and supramolecular levels and to quantify the effect of ion concentration, ion valance, pH, and temperature on the structure and macroscopic (thermodynamic) properties of the tissue. We also apply computational techniques to model water-ion-biopolymer interactions in these systems.
A major success of this basic polymer physics approach has been to exploit our understanding of water-polymer interactions to tailor the properties of diffusion MRI phantoms so as to calibrate MRI scanners, specifically to assure quality of the imaging data and to assess scanner performance on an on-going basis. Our recently issued U.S. Patent for a “Phantom for diffusion MRI imaging” is allowing, for the first time, quantitative diffusion MRI studies to be performed. The polymer polyvinylpyrrolidone (PVP) has ideal properties for this demanding application. It is chemically and thermally stable, has a long shelf life, is safe and non-toxic, can be shipped from site to site, and has stable diffusion and relaxation properties. Michael Boss, NIST has incorporated our PVP polymer in NIST’s own diffusion MRI standard. The technology is also being promulgated commercially e.g., by https://hpd-online.com/more-hpd-products/.
Functional properties of extracellular matrix (ECM)
Our goal is to understand and quantify the interactions among the major macromolecular components of ECM that give rise to its unique functional properties. ECM is present in every tissue and performs a key role in determining normal and abnormal organ function. Specifically, we are studying interactions among the primary ECM components, namely collagen, proteoglycans (PG), water, and ions, which govern ECM’s macroscopic biomechanical and transport properties, using cartilage as a model system. The biomechanical behavior of cartilage and other ECMs reflects biochemical and microstructural changes occurring during development, disease, degeneration, and aging. Understanding the basis of important functional properties of cartilage, particularly its load-bearing and lubricating abilities, requires an array of experimental techniques that probe a wide range of relevant length and time scales. Understanding the physical and chemical mechanisms affecting cartilage swelling (hydration) is essential for predicting these properties, which are mainly governed by osmotic and electrostatic forces. This knowledge can inform tissue-engineering or regenerative-medicine strategies to grow, repair, and reintegrate replacement cartilage. To obtain a self-consistent physical picture of tissue structure/function relationships, we measure various physical/chemical properties of ECM tissues and tissue analogs at different length- and time-scales using a variety of complementary static and dynamic experimental techniques, e.g., osmometry, SANS, SAXS, neutron spin-echo (NSE), SLS, DLS, AFM, and fluorescence correlation spectroscopy (FCS).
Controlled tissue hydration provides a direct means of determining the viability and load-bearing ability of cartilage. Previously, we designed and built a tissue micro-osmometer to perform high-precision swelling pressure measurements on small tissue samples (less than 1 microgram) as a function of the water activity (vapor pressure). We make osmotic pressure measurements to determine how the individual components of cartilage ECM (e.g., aggrecan, hyaluronic acid [HA], and collagen) contribute to the total load-bearing capacity of the tissue. We demonstrated that aggrecan–HA aggregates self-assemble into microgels, contributing to improved dimensional stability and the tissue’s lubricating ability. We also found that aggrecan is highly insensitive to changes in the ionic environment, particularly to the concentration of calcium ions, which is critically important in maintaining the tissue’s mechanical integrity in high Ca2+ environments and allowing aggrecan to serve as a calcium ion reservoir in cartilage and bone.
We recently developed a new biomimetic model of cartilage ECM consisting of a dispersion of polyacrylic acid (PAA) microgel particles embedded within a polyvinyl alcohol (PVA) gel matrix. In this system, PAA mimics the behavior of proteoglycan assemblies while PVA mimics the role of the collagen network. The PVA/PAA biomimetic model system reproduces not only the shape of the cartilage swelling pressure curves, but also the numerical values reported for healthy and osteoarthritic human cartilage samples. Systematic studies made on model composite hydrogels is expected to provide invaluable insights into the effects of various factors (matrix stiffness, swelling pressure, fixed charge density, synovial fluid composition, etc.) on the macroscopic mechanical/swelling properties, and ultimately the load-bearing ability of cartilage. Similar systematic studies cannot be obtained from measurements made on biological tissues because their composition and physical properties cannot be independently and systematically varied as they can in these synthetic model polymer composites.
The resistance of tissue to external loads is determined by its osmotic modulus. Therefore, maps of the osmotic modulus are particularly useful for characterizing the load-bearing properties of cartilage. We developed a method that utilizes the precise scanning capabilities of the AFM to generate compliance maps, from which relevant elastic properties can be extracted. We then combined AFM with tissue micro-osmometry to generate elastic and osmotic modulus maps of cartilage.
We have begun translating this critical tissue-science understanding of the structure/function relationships of components of ECM to develop and design novel non-invasive MR imaging methods with the aim of inferring ECM composition, patency, and functional properties in vivo. Our goal is to use MRI for early diagnosis of cartilage and other ECM diseases, as well as to provide a means for following normal and abnormal development of the ECM. This challenging project entails making ‘invisible’ components of ECM, (e.g., collagen and PGs) ‘visible’ and then using our understanding of biopolymer interactions to predict tissue load-bearing properties. One major obstacle is that water molecules bound to immobile species (e.g., collagen) cannot be visualized with conventional MRI approach. However, magnetization transfer (MT) MRI (as well as other methods) make it possible to detect these bound protons indirectly by transferring their magnetization to the free water surrounding them. It also makes it possible to estimate the collagen content in tissue. In a pilot study, we applied the new MT MRI method to determine the concentration and distribution of the main macromolecular constituents in bovine femoral-head cartilage samples. The results obtained by the MT MRI method were qualitatively consistent with those obtained by histological techniques, such as high-definition infrared (HDIR) spectroscopy. Our work was aided by the previous receipt of a DIR Director’s Award to investigate this proposed line of research, along with our collaborators Sergey Leikin and Edward Mertz.
Additional Funding
- Award 305500-1.01-60855 from the Henry Jackson Foundation supports the Section's project in “Enhanced Software Tools for the Analysis of Diffusion MRI in TBI and PTSD,” which is under the joint auspices of the NIH, DoD, CNRM, and USUHS.
- Award 306135-2.01-60855 from the Henry Jackson Foundation supports the Section's project “Clinical Double Pulsed-Field Gradient (dPFG) MRI for Mild TBI Assessment,” which is under the joint auspices of the NIH, DoD, CNRM, and USUHS.
Publications
- Bai R, Stewart C, Plenz D, Basser PJ. Assessing the sensitivity of diffusion MRI to detect neuronal activity directly. Proc Natl Acad Sci USA 2016 113(12):E1728-E1737.
- Horkay F, Basser PJ. Osmotic properties of cartilage. In: Xia Y, Momot K, eds. Biophysics and Biochemistry of Cartilage by NMR and MRI. Royal Soc Chem 2016 44-61.
- Benjamini D, Basser PJ. Use of marginal distributions constrained optimization (MADCO) for accelerated 2D MRI relaxometry and diffusometry. J Magn Reson 2016 271:40-45.
- Horkay F, Pierpaoli C, Basser PJ. Phantom for diffusion MRI imaging. U.S. Patent 9,603,546, filed January 26, 2010; issued March 28, 2017.
- Arrigoni F, Peruzzo D, Gagliardi C, Maghini C, Colombo P, Iammarrone FS, Pierpaoli C, Triulzi F, Turconi AC. Whole-brain DTI assessment of white matter damage in children with bilateral cerebral palsy: evidence of involvement beyond the primary target of the anoxic insult. AJNR Am J Neuroradiol 2016 37(7):1347-1353.
Collaborators
- Filippo Arrigoni, MD, Fondazione IRCCS Eugenio Medea, Bosisio Parini, Italy
- Madison Berl, PhD, Children's National Medical Center, Washington, DC
- Emilios Dimitriadis, PhD, Division of Bioengineering and Physical Science, NIBIB, Bethesda, MD
- Uzi Eliav, PhD, Tel Aviv University, Tel Aviv, Israel
- Erik Geissler, PhD, CNRS, Université Joseph Fourier de Grenoble, Grenoble, France
- Iren Horkayne-Szakaly, MD, Armed Forces Institute of Pathology, Washington, DC
- Sharon Juliano, PhD, Uniformed Services University of the Health Sciences, Bethesda, MD
- Sergey Leikin, PhD, Section on Physical Biochemistry, NICHD, Bethesda, MD
- Stefano Marenco, PhD, Clinical Brain Disorders Branch, NIMH, Bethesda, MD
- Edward L. Mertz, PhD, Section on Physical Biochemistry, NICHD, Bethesda, MD
- Pedro Miranda, PhD, Universidade de Lisboa, Lisbon, Portugal
- Gil Navon, PhD, Tel Aviv University, Tel Aviv, Israel
- Uri Nevo, PhD, Tel Aviv University, Tel Aviv, Israel
- Evren Özarslan, PhD, Brigham and Women's Hospital, Boston, MA
- Sinisa Pajevic, PhD, Mathematical and Statistical Computing Laboratory, CIT, NIH, Bethesda, MD
- Bradley Roth, PhD, Oakland University, Rochester, MI
- Susan Swedo, MD, Pediatrics and Developmental Neuroscience Branch, NIMH, Bethesda, MD
- Lindsay Walker, MSc, Brown University, Providence, RI
- Katherine Warren, MD, Pediatric Oncology Branch, Center for Cancer Research, NCI, Bethesda, MD
- Brain Development Cooperative Group, Various
Contact
For more information, email pjbasser@helix.nih.gov or visit sqits.nichd.nih.gov.


