The Arms Race between KRAB-ZFPs and Transposable Elements and its Impact on Mammals

- Todd S. Macfarlan, PhD, Head, Section on Section on Mammalian Development and Evolution
- Sherry Ralls, BA, Biologist
- Jinpu Jin, PhD, Visiting Fellow
- Mohamed Mohamed, PhD, Visiting Fellow
- Ming-an Sun, PhD, Visiting Fellow
- Gernot Wolf, PhD, Visiting Fellow
- Peng Yang, PhD, Visiting Fellow
- Michael Evans, DO, Clinical Fellow
- Justin Demmerle, BA, Graduate Student
- Alyssa Kosturakis, MSc, Medical Research Scholars Program Fellow
- Drew Honson, BS, Postbaccalaureate Fellow
- Sienna Schaeffer, BS, Postbaccalaureate Fellow
- Guillaume Furey, Intern
- Elaine Wu, Intern
At the NICHD, our central mission is to ensure that every human is born healthy. Despite much progress in understanding the many ways the mother interacts with the fetus during development, we still know little about the molecular changes that promoted the emergence of placental mammals over 100 million years ago from our egg-laying relatives, nor those mechanisms that continue drive phenotypic differences amongst mammals. One attractive hypothesis is that retroviruses and their endogenization into the genomes of our ancestors played an important role in eutherian evolution, by providing protein coding genes such as syncytins (derived from retroviral env genes that cause cell fusions in placental trophoblasts) and novel gene-regulatory nodes that altered expression networks to allow for implantation and the emergence and continued evolution of the placenta. Our primary interest is to explore the impact of these endogenous retroviruses (ERVs), which account for about 10% of our genomic DNA, on embryonic development and on the evolution of new traits in mammals. This has led us to examine the rapidly evolving Kruppel-associated box zinc-finger protein (KZFP) family, the single largest family of transcription factors (TFs) in most, if not all, mammalian genomes. Our hypothesis is that KZFP gene expansion and diversification was driven primarily by the constant onslaught of ERVs and other transposable elements (TEs) to the genomes of our ancestors, as a means to transcriptionally repress them. The hypothesis is supported by recent evidence demonstrating that the majority of KZFPs bind to TEs and that TEs and nearby genes are activated in KZFP knockout mice. We will continue to explore the impacts of the TE/KZFP “arms race” on the evolution of mammals. We will also begin a new phase exploring whether KZFPs play broader roles in genome regulation, beyond gene silencing, and how these functions impact mammalian development.
Kruppel-associated box zinc-finger proteins (KRAB-ZFPs) have emerged as candidates that recognize ERVs. KRAB-ZFPs are rapidly evolving transcriptional repressors that emerged in a common ancestor of coelacanth, birds, and tetrapods. They make up the largest family of transcription factors in mammals (estimated to be several hundred in mice and humans). Each species has its own unique repertoire of KRAB-ZFPs, with a small number shared by closely related species and a larger fraction specific to each species. Despite their abundance, little is known about their physiological functions. KRAB-ZFPs consist of an N-terminal KRAB domain that binds to the co-repressor KAP1 and a variable number of C-terminal C2H2 zinc finger domains that mediate sequence-specific DNA binding. KAP1 directly interacts with the KRAB domain, which recruits the histone methyltransferase (HMT) SETDB1 and heterochromatin protein 1 (HP1) to initiate heterochromatic silencing. Several lines of evidence point to a role for the KRAB-ZFP family in ERV silencing. First, the number of C2H2 zinc finger genes in mammals correlates with the number of ERVs. Second, the KRAB-ZFP protein ZFP809 was isolated based on its ability to bind to the primer binding site for proline tRNA (PBSpro) of murine leukemia virus (MuLV). Third, deletion of the KRAB-ZFP co-repressors Trim28or Setdb1 leads to activation of many ERVs. We have therefore begun a systematic interrogation of KRAB-ZFP function as a potential adaptive repression system against ERVs.
We focused on ZFP809 as a likely ERV–suppressing KRAB-ZFP, given that it was originally identified as part of a repression complex that recognizes infectious MuLV via direct binding to the 18 nt Primer Binding Site for Proline (PBSpro) sequence. We hypothesized that ZFP809 might function in vivo to repress other ERVs that utilized the PBSpro. Using ChIP-seq of epitope-tagged ZFP809 in embryonic stems cells (ESCs) and embryonic carcinoma (EC) cells, we determined that ZFP809 binds to several sub-classes of ERV elements via the PBSpro. We generated Zfp809 knockout mice to determine whether ZFP809 was required for silencing the ERV element VL30pro. We found that Zfp809 knockout tissues displayed high levels of VL30pro elements and that the targeted elements display an epigenetic shift from repressive epigenetic marks (H3K9me3 and CpG methylation) to active marks (H3K9Ac and CpG hypo-methylation). ZFP809–mediated repression extended to a handful of genes that contained adjacent VL30pro integrations. Furthermore, using a combination of conditional alleles and rescue experiments, we determined that ZFP809 activity was required in development to initiate silencing, but not in somatic cells to maintain silencing. These studies provided the first demonstration for the in vivo requirement of a KRAB-ZFP in the recognition and silencing of ERVs.
As a follow-up to our studies on ZFP809, we have begun a systematic analysis of KRAB-ZFPs using a medium-throughput ChIP-seq screen and functional genomics of KRAB-ZFP clusters and individual KRAB-ZFP genes. Our ChIP-seq data demonstrate that the majority of recently evolved KRAB-ZFP genes interact with and repress distinct and partially overlapping ERV and other retrotransposons targets. The hypothesis is strongly supported by the distinct ERV reactivation phenotypes we observed in mouse ESC lines lacking one of five of the largest KRAB-ZFP gene clusters. Furthermore, our preliminary evidence suggests that KRAB-ZFP cluster KO mice are viable, but have elevated rates of somatic retrotransposition of specific retrotransposon families, providing the first direct genetic link between KRAB-ZFP gene diversification and retrotransposon mobility.
Click image to view.
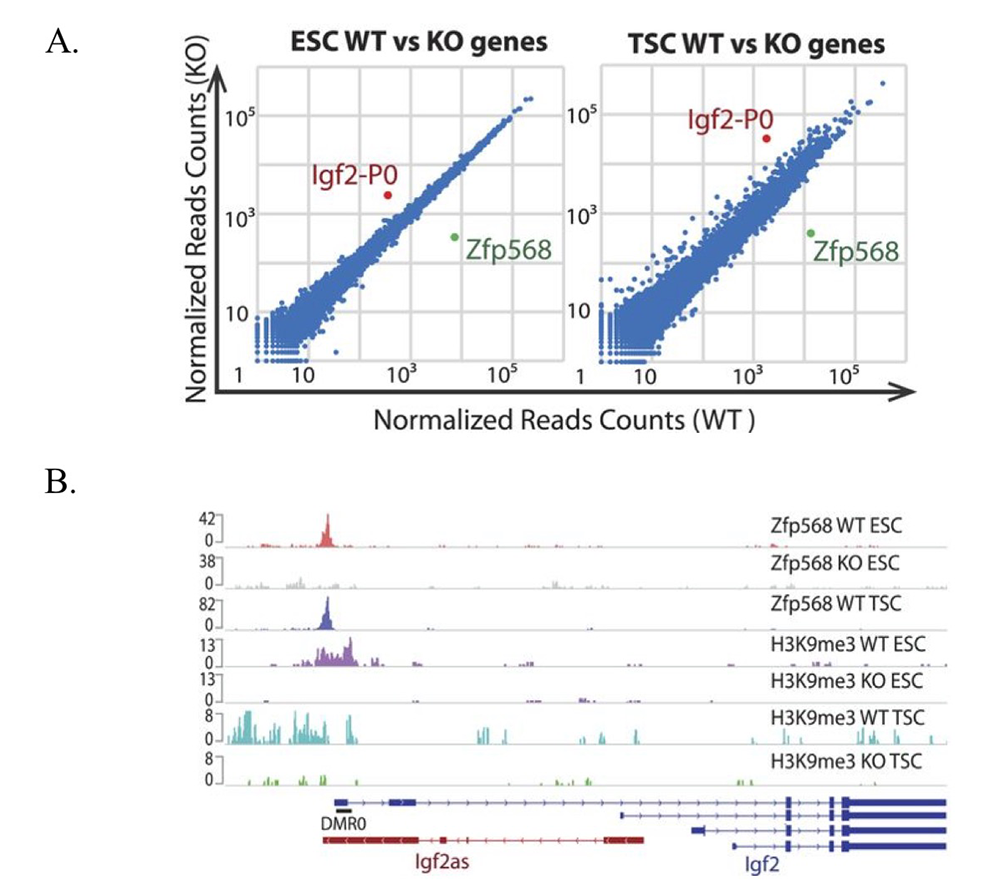
Figure 1. ZFP568 directly represses the Igf2-P0 transcript.
A. Scatter plots of gene expression in Zfp568 wild-type (WT) and knockout (KO) embryonic stem cells (ESCs) and trophoblast stem cells (TSCs), as determined by RNA-seq.
B. ZFP568 and H3K9me3 ChIP-seq signals at the Igf2 locus in Zfp568 WT and KO ESCs and TSCs. DMR0 is a differentially methylated region overlapping exon 1 of the Igf2-P0 transcript. Igf2as is the Igf2 antisense transcript.
Although our data show that many KRAB-ZFPs repress ERVs, we also found that more ancient KRAB-ZFPs, which emerged in a human/mouse common ancestor, do not bind or repress ERVs. One of these KRAB-ZFPs, ZFP568, plays an important role in silencing a key developmental gene that may have played a critical role in the onset of viviparity in mammals. Using ChIP-seq and biochemical assays, we determined that ZFP568 is a direct repressor of a placental-specific isoform of the Igf2 gene called Igf2-P0 (Figure 1). Insulin-like growth factor 2 (Igf2) is the major fetal growth hormone in mammals. We demonstrated that loss of Zfp568, which causes gastrulation failure, or mutation of the ZFP568 binding site at the Igf2-P0 promoter, cause inappropriate Igf2-P0 activation. We also showed that this lethality could be rescued by deletion of Igf2. The data highlight the exquisite selectivity by which members of the KRAB-ZFP family repress their targets and identify an additional layer of transcriptional control of a key growth factor regulating fetal and placental development. In an exciting follow-up to these studies, we determined that ZFP568 is highly conserved and under purifying selection in eutheria with the exception of human. Human ZNF568 allele variants have lost the ability to bind to and repress Igf2-P0, which may have been driven by the loss of the Igf2-p0 transcript in human placenta. We solve the crystal structure of mouse ZFP568 zinc fingers bound to the Igf2-P0 binding site, which reveals several non-canonical ZF-DNA contacts, highlighting the ability of individual ZFs to change confirmation depending upon ZF context and DNA structure (Figure 2). The structures also explain how mutations in human ZNF568 alleles disrupt Igf2-P0 interactions, which contain either deleted ZFs or mutations of key ZF-DNA contact residues. Taken together, our studies provide important insights into the evolutionary and structural dynamics of ZF-DNA interactions, which play a key role in regulating mammalian development and evolution.
Click image to view.
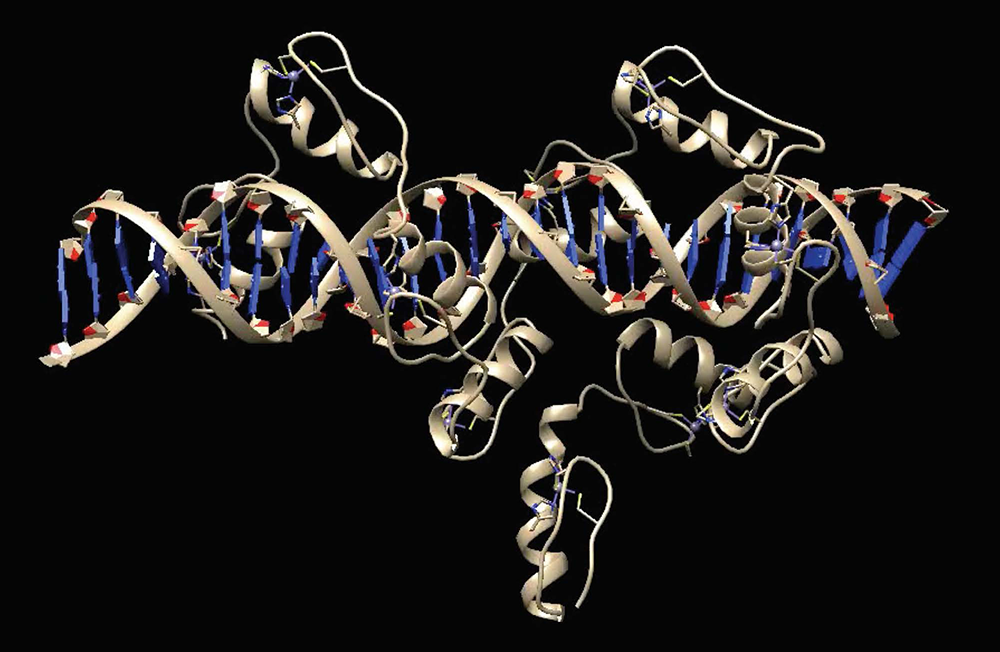
Figure 2. Structure of ZFP568 zinc fingers bound to the Igf2-P0 target sequence
ZFP568 uses zinc fingers 3–11 to achieve high-affinity binding to the Igf2-P0 target sequence. An A-T–rich stretch in the target sequence alters the minor grove distance, leading to non-canonical interactions between zinc fingers and DNA, highlighting the flexibility of zinc fingers in DNA–binding modes to accommodate altered DNA structures.
Additional Funding
- Director's Award, The role of retrotransposons and zinc finger proteins in neural development and disease, Co-PI Levin, on-going
- Human Placenta Project, Exploring KRAB-ZFP gene function in placental development
Publications
- Patel A1, Yang P1, Tinkham M, Pradham M, Sun MA, Wang Y, Hoang D, Wolf G, Horton JR, Zhang X, Macfarlan T, Cheng X. DNA conformation induces adaptable binding by tandem zinc finger proteins. Cell 2018;Mar 22;173:221-233.
- Yang P, Wang Y, Hoang D, Tinkham M, Patel A, Sun M-A, Wolf G, Baker M, Chien H-C, Lai N, Cheng X, Shen C-K J, Macfarlan TS. A placental growth factor is silenced in mouse embryos by the zinc finger protein ZFP568. Science 2017;356 (6339):757-759.
- Wolf G, Rebollo R, Karimi MM, Ewing AD, Kamada R, Wu W, Wu B, Bachu M, Ozato K, Faulkner GJ, Mager DL, Lorincz MC, Macfarlan TS. On the role of H3.3 in retroviral silencing. Nature 2017;548(7665):E1-E3.
- Dunn-Fletcher CE, Muglia LM, Pavlicev M, Wolf G, Sun MA, Hu YC, Huffman E, Tumukuntala S, Thiele K, Mukherjee A, Zoubovsky S, Zhang X, Swaggart KA, Lamm KYB, Jones H, Macfarlan TS, Muglia LJ. Anthropoid primate-specific retroviral element THE1B controls expression of CRH in placenta and alters gestation length. PLoS Biol 2018;16(9):e2006337.
- Honson DD, Macfarlan TS. A lncRNA-like role for LINEs in development. Dev Cell 2018;46:132-134.
Collaborators
- Xiaodong Cheng, PhD, Emory University, Atlanta, GA
Contact
For more information, email todd.macfarlan@nih.gov or visit http://macfarlan.nichd.nih.gov.


