Organ and Tissue Formation during Development

- Brant M. Weinstein, PhD, Head, Section on Vertebrate Organogenesis
- Aniket Gore, PhD, Staff Scientist
- Van Pham, BS, Scientific Technician
- Hyun Min Jung, PhD, Postdoctoral Fellow
- Mayumi Miller, PhD, Postdoctoral Fellow
- Scott Paulissen, PhD, Postdoctoral Fellow
- Laura Pillay, PhD, Postdoctoral Fellow
- Amber Stratman, PhD, Postdoctoral Fellow
- Kiyohito Taimatsu, PhD, Postdoctoral Fellow
- Marina Venero Galanternik, PhD, Postdoctoral Fellow
- Alexandra Fister, BS, Postbaccalaureate Fellow
- Ryan Gober, BS, Postbaccalaureate Fellow
- Avery Swearer, BS, Postbaccalaureate Fellow
- Joseph Yano, BS, Postbaccalaureate Fellow
The major focus of the Section is to understand how the elaborate networks of blood and lymphatic vessels arise during vertebrate development. Blood vessels supply every tissue and organ with oxygen, nutrients, and cellular and humoral factors. Lymphatic vessels drain fluids and macromolecules from the interstitial spaces of tissues, returning them to the blood circulation, and they play an important role in immune responses. Our studies on the formation of blood and lymphatic vessels are of great clinical interest because of the roles both types of vessels play in cancer and ischemia.
The zebrafish (Danio rerio) is a small tropical freshwater fish that possesses a unique combination of features that make it particularly suitable for studying vessel formation. Zebrafish are genetically tractable vertebrates with externally developing, optically clear embryos that are readily available for observation and experimental manipulation. Such features permit observation of every vessel in the living animal and simple, rapid screening for even subtle vascular-specific defects. Our current studies use genetic screening, experimental analysis, and imaging to examine cues directing vascular patterning and morphogenesis, regulation of vascular integrity, assembly of the lymphatic system, and the roles of novel vascular-associated cells.
In addition to our work on vessel development, we are also pursuing studies to understand the role of epigenetics during early development, in particular how DNA methylation and other epigenetic mechanisms help to coordinate cell, tissue, and organ specification and differentiation.
Specification and patterning of developing blood vessels
We are working to elucidate the cellular and molecular mechanisms responsible for the specification, patterning, and differentiation of blood vessels during development. Blood vessels are ubiquitous and vital components of vertebrate animals, innervating and supplying every tissue and organ with oxygen and nutrients. Many of the recent insights into mechanisms of blood vessel formation have come from studies in model organisms including the zebrafish. In zebrafish every blood vessel can be observed in living animals with high resolution, and simple, rapid screening can be accomplished for even subtle vascular-specific mutants (Figure 1). We are carrying out a several related projects using the fish, including:
- New tools for experimental analysis of vascular development. This includes generating novel transgenic lines for visualizing different endothelial cell and perivascular cell types and for driving gene expression or performing molecular profiling of mRNAs and microRNAs in these same cell populations.
- Genetic analysis of vascular development. We have identified many novel mutants affecting vascular development in our transgene-assisted forward genetic screens, and we are currently characterizing the phenotypes and molecular basis for a number of these mutants.
- Analysis of vascular specification, patterning, and morphogenesis. We are studying the development of several vascular beds including the vasculature of the pectoral fin, the fish equivalent of the mammalian forelimb.
Click image to view.
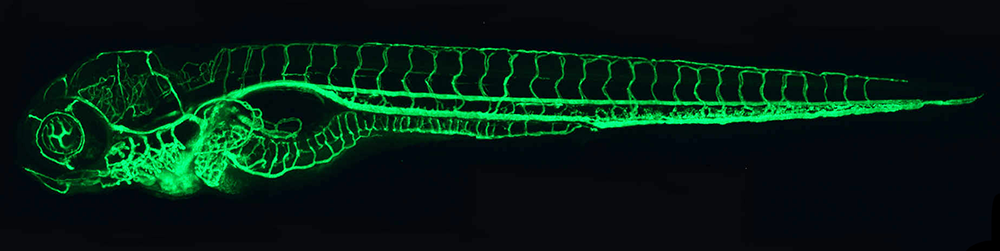
Figure 1. The zebrafish vascular system
Confocal micro-angiogram of the vascular system of a 4½-day-old zebrafish larva labeled by injecting fluorescent microspheres. The transparency of zebrafish larvae makes it possible to use high-resolution optical imaging methods to visualize the entire vasculature in exquisite detail.
Specification and patterning of the lymphatic system
The lymphatic system is a completely separate vascular system from the blood circulatory system and comprises an elaborate blind-ended tree of vessels that extensively innervate most of the body, emptying lymph fluid into the venous blood vascular system via several evolutionarily conserved drainage points. The lymphatic system is essential for immune responses, fluid homeostasis, and fat absorption, and is involved in many pathological processes, including tumor metastasis and lymphedema. However, progress in understanding the origins and early development of the system has been hampered by difficulties in observing lymphatic cells in vivo and performing defined genetic and experimental manipulation of the lymphatic system in currently available model organisms. Our groundbreaking studies demonstrated that zebrafish possess a lymphatic system that shares many of the morphological, molecular, and functional characteristics of lymphatic vessels found in other vertebrates, providing a powerful model for the purpose of imaging and studying lymphatic development. We are currently pursuing further study of the formation of the lymphatic system through several ongoing projects:
- We generated new transgenic lines that permit direct, specific visualization of the developing lymphatic vasculature and are using sophisticated imaging of these transgenic animals to characterize lymphatic development (Figure 2).
- We carried out forward-genetic ENU (N-ethyl-N-nitrosourea) mutagenesis screens using our lymphatic reporter transgenic lines to identify new lymphatic-specific mutants with defects in novel genes that play important roles in lymphatic development.
- We are characterizing and studying novel micro RNAs expressed in the lymphatic endothelium and how these small regulatory RNAs influence lymphatic gene expression and lymphatic development.
The results of our studies, combining the genetic and experimental tools available in the zebrafish with the ability to perform high-resolution microscopic imaging of developing vascular structures in living animals, will continue to lead to important new insights into the origins and growth of the lymphatic system and molecular mechanisms that are critical in lymphatic development and lymphatic pathologies.
Click image to view.
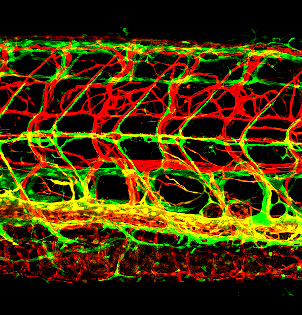
Figure 2. Novel lymphatic vascular reporter
Lateral view confocal image of the trunk of a 12 dpf (days post-fertilization) Tg(kdrl:cherry); Tg(mrc1a:egfp) double-transgenic zebrafish with red fluorescent blood vessels and green fluorescent lymphatics. See Reference 1 for more information.
Regulation of vascular integrity
We are using the zebrafish to understand the cellular and molecular mechanisms responsible for proper vessel morphogenesis and for the generation and maintenance of vascular integrity. Disruption of vascular integrity is associated with hemorrhagic stroke, a severe and debilitating form of stroke associated with high morbidity and mortality. Meningeal vascular dysfunction is also associated with a neurocognitive deficits and neurodegenerative disease. Many of the recent insights into molecular mechanisms regulating vascular integrity have come from studies in model organisms such as the zebrafish. We are pursuing several related projects.
Click image to view.
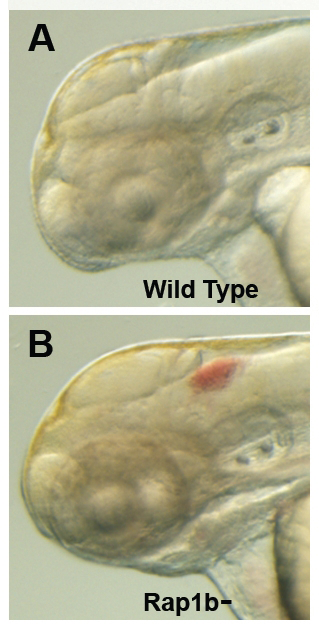
Figure 3. Intracranial hemorrhage (ICH) in the developing zebrafish
The clarity of zebrafish larvae also makes it straightforward to screen for animals with intracranial hemorrhage, as is evident in comparing lateral views of a 2-day-old wild-type larva (A) with a hemorrhage-prone larva deficient in rap1b (B).
- Genes regulating vascular integrity. We used forward-genetic screens to identify new zebrafish mutants that disrupt cranial vascular integrity in the zebrafish (Figure 3), using exome sequencing and SNP (single nucleotide polymorphism) analysis with a newly developed SNP database to perform higher-throughput cloning of mutants. We already characterized the role of GDF6 (growth differentiation factor 6, also known as BMP13) in vascular integrity, demonstrating that the gene promotes maintenance of vascular integrity by suppressing excess VEGF (vascular endothelial growth factor) signaling. We are currently characterizing the molecular nature of the defects in several additional mutants.
- Acquisition and function of supporting vascular smooth muscle cells. The vascular smooth cells (VSMC) that surround the endothelial tube play a critical role in regulating vascular tone and vascular integrity. We examined the early origins of these cells, how the interaction of these cells with endothelial tubes helps maintain the vascular basement membrane and restricts vessel diameter, and the molecular mechanisms underlying the arterial (versus venous) specific recruitment of VSMC.
- Vasculature and vascular-associated cells in the meninges. The meninges are an external enveloping connective tissue that encases the brain, producing cerebrospinal fluid, acting as a cushion against trauma, nourishing the brain via nutrient circulation, and removing waste. Despite its importance, the cell types present in the meninges and the function and embryonic origins of this tissue are still not well understood. We recently discovered and characterized fluorescent granular perithelial cells (FGPs) in the zebrafish, a novel endothelium-derived perivascular cell population closely associated with meningeal blood vessels that is likely to have a critical role in meningeal function (Figure 4). We are currently carrying out several different studies to understand the function of these and other novel meningeal vascular-associated cell populations.
Click image to view.
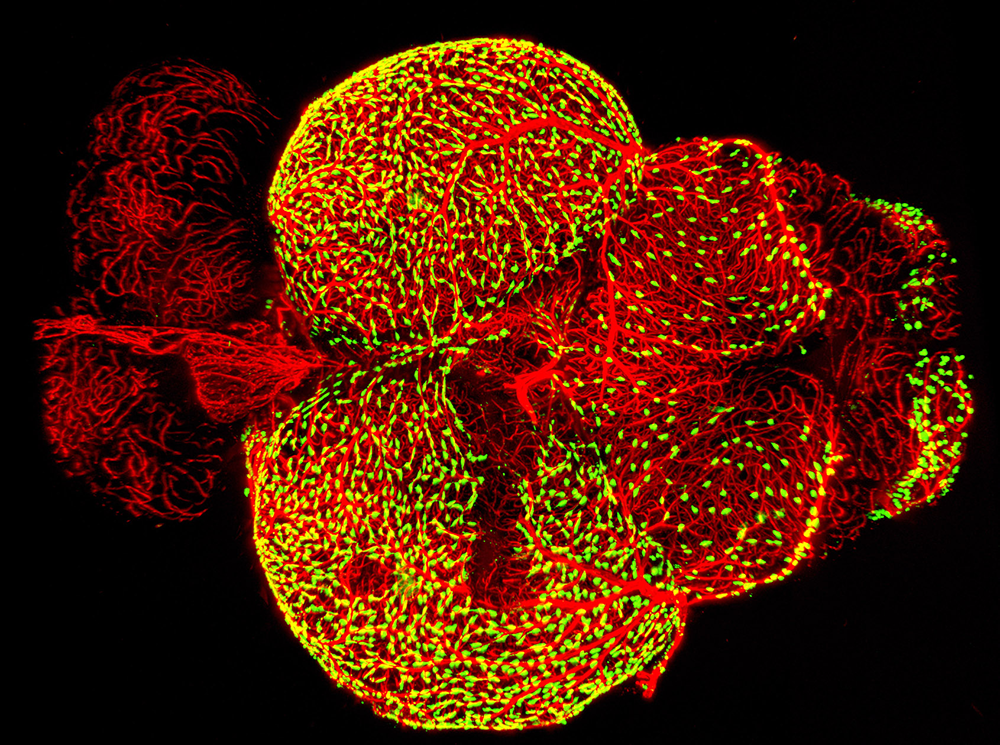
Figure 4. Novel perivascular cells on the zebrafish brain
Confocal micrograph of "Fluorescent Granular Perithelial Cells" (FGPs, in green) adhering to the outside of meningeal blood vessels (in purple) on the brain of a Tg(mrc1a:egfp); Tg(kdrl:cherry) double-transgenic adult zebrafish. We recently showed that FGPs are unique endothelium-derived perivascular cells with unusual scavenging properties that are likely critical for brain homeostasis. See Reference 4 for additional details.
Epigenetics of development
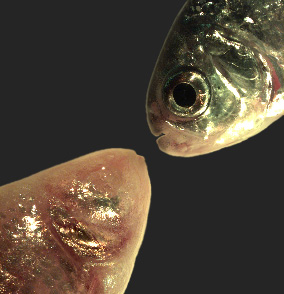
Click image to view.
Figure 5. Mexican tetra cave and surface fish
The Mexican tetra Astyanax mexicanus is a freshwater fish native to parts of southern Texas and eastern and central Mexico, which exists in both surface-dwelling ("surface morphs," top right) and very closely related cave-dwelling ("cave morphs," bottom left) populations. Cave morphs have a series of uniquely evolved adaptations including loss of eyes and pigment, dramatically altered metabolism, altered vascular function, and altered sleep regulation and behavior. Results from our laboratory suggest that altered DNA methylation and resulting coordinated changes in expression of large sets of genes have helped to drive at least some of this rapid evolutionary change. See Reference 5 for details.
We are using the genetically and experimentally accessible zebrafish and Mexican tetra (Astyanax mexicanus) models to uncover the molecular basis for organ- and tissue-specific epigenetic regulation during development in the following interrelated projects:
- Role of the DNA methylase Dnmt3bb.1 in hematopoiesis and eye development. DNA methyltransferases (DNMTs) are responsible for placing methyl marks on DNA. Promoter DNA methylation is typically associated with gene repression, while gene body methylation is generally associated with activation of a gene locus. We showed that Dnmt3bb.1 mediates hematopoietic stem and progenitor cell (HSPC) maintenance by methylating the gene body region of the cmyb locus, leading to the sustained expression of this essential transcription factor for maintaining hematopoietic specification. More recently, we showed that the same DNA methyltransferase mediates global repression of eye development genes, and that excess DNMT3B expression promotes eye loss in blind eyeless Astyanax cavefish (Figure 5).
- Epigenetic regulation of metabolism using Astyanax cavefish. The fish have extreme and unusual metabolic adaptations that allow them to survive chronic and long-term food deprivation. We are using molecular profiling and other methods to examine changes in the regulation of metabolic genes and the role of epigenetics in these changes.
- Forward genetic screen for epigenetic regulatory factors. Genetic screens carried out in Drosophila and C. elegans have been highly successful in identifying genes regulating cell-type specific epigenetic gene regulation in invertebrates, but the molecular mechanisms involved in organ- and tissue-specific epigenetic regulation in vertebrates are still relatively unknown. We developed a novel zebrafish transgenic reporter line that allows us to monitor dynamic changes in epigenetic regulation in intact animals during development. Using this transgenic line, we are performing the first large-scale F3 genetic screen in a vertebrate to identify recessive mutants in regulators of epigenetic gene silencing or activation (Figure 6).

Click image to view.
Figure 6. An epigenetic silencing mutant in the zebrafish
Lateral views of the head and anterior trunk of a wild type (top) and tissue-specific epigenetic silencing mutant (bottom) zebrafish. The mutant causes loss of epigenetic silencing specifically in the liver (red arrows), as visualized with a novel transgenic reporter line developed in our lab that permits dynamic, tissue-specific visualization of epigenetic silencing in living animals.
Publications
- Jung H-M, Castranova D, Swift MR, Pham VN, Galanternik MV, Isogai S, Butler MG, Mulligan TS, Weinstein BM. Development of the larval lymphatic system in the zebrafish. Development 2017;144:2070-2081.
- Krispin S, Melick CH, Stratman AN, Malinverno M, Stan RV, Gleklen J, Castranova D, Dejana E, Weinstein, BM. Growth Differentiation Factor 6 (GDF6) promotes vascular stability by counteracting VEGF activity. Arterioscler Thromb Vasc Biol 2018;38:353-362.
- Stratman AN, Pezoa SA, Farrelly OM, Castranova D, Dye LE, Sidik H, Talbot WS, Weinstein BM. Mural-endothelial cell-cell interactions stabilize the developing zebrafish dorsal aorta. Development 2017;144:115-127.
- Venero Galanternik M, Castranova D, Gore AV, Jung HM, Stratman AN, Miller MF, Diemer J, Blewett N, Pham VN, Marquart G, Burgess H, Maraia R, Liu PP, Weinstein BM. A novel perivascular cell in the zebrafish brain. eLife 2017;6:e24369.
- Gore AV, Tomins KA, Iben J, Ma L, Castranova D, Davis AE, Parkhurst A, Jeffery WR, Weinstein BM. An epigenetic mechanism for cavefish eye degeneration. Nat Ecol Evol 2018;2:1155-1160.
Collaborators
- Andreas Baxevanis, PhD, Computational and Statistical Genomics Branch, NHGRI, Bethesda, MD
- Harold Burgess, PhD, Section on Behavioral Neurogenetics, NICHD, Bethesda, MD
- George Davis, PhD, University of Missouri-Columbia, Columbia, MO
- Elisabetta Dejana, PhD, The FIRC Institute of Molecular Oncology Foundation, Milan, Italy
- Silvio Gutkind, PhD, Oral and Pharyngeal Cancer Branch, NIDCR, Bethesda, MD
- James Iben, PhD, Molecular Genomics Laboratory, NICHD, Bethesda, MD
- Sumio Isogai, PhD, Iwate Medical University, Morioka, Japan
- William R. Jeffery, PhD, University of Maryland, College Park, MD
- Paul Liu, MD, PhD, Genetics and Molecular Biology Branch, NHGRI, Bethesda, MD
- Richard Maraia, MD, Section on Molecular and Cell Biology, NICHD, Bethesda, MD
- Yoh-suke Mukouyama, PhD, Laboratory of Stem Cell and Neuro-Vascular Biology, NHLBI, Bethesda, MD
- Lisa M. Price, PhD, Division of Developmental Biology, NICHD, Bethesda, MD
- Radu V. Stan, MD, PhD, Geisel School of Medicine at Dartmouth, Lebanon, NH
Contact
For more information, email weinsteb@mail.nih.gov or visit http://uvo.nichd.nih.gov.


